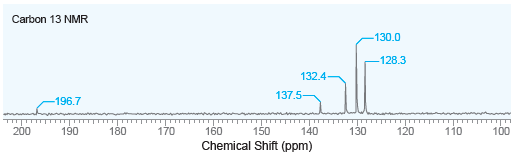
A compound with molecular formula C 13 H 10 O produces a strong signal at 1660 cm
Question:
Transcribed Image Text:
Carbon 13 NMR -130.0 -128.3 132.4- 137.5- -196.7 200 180 170 160 150 Chemical Shift (ppm) 140 130 120 110 100 190
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C 9 H 10 O exhibits the following spectra ( 1 H NMR, 13 C NMR, and IR). Identify the structure of this compound.. Proton NMR 10 Chemical Shift (ppm) Carbon NMR 128.5...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
A compound with molecular formula C 8 H 10 O produces six signals in its 13 C NMR spectrum and exhibits the following 1 H NMR spectrum. Deduce the structure of the compound. Proton NMR Chemical Shift...
-
The table shows the fees for refund anticipation loans (RALs) offered by an online tax preparation firm. Find the annual rate of interest for each of the following loans. Assume a 360-day year. (A) A...
-
Pete is considering placing a bet on the NCAA playoff game between Indiana and Purdue. Without any further information, he believes that each team has an equal chance to win. If he wins the bet, he...
-
Relating Graphs: For the oscillator DE + 0.25x = 0, Fig. 4.1.4 shown previously linked solution graphs and phase portrait. Parts (a), (b), (c), and (d) relate to that figure. (a) Mark on the phase...
-
Explain in a few lines why diversifiable risk cannot be remunerated on markets in equilibrium?
-
Presented below are selected ledger accounts of Vu Corporation at December 31, 2014. Cash .................. $ 92,500 Merchandise inventory ........... 267,500 Sales .................. 2,137,500...
-
Basics of Productivity Measurement Holbrook Company gathered the following data for the past two years: Base Year Current Year Output 930,000 1,050,000 Output prices $14 $14 Input quantities:...
-
Financial information for American Eagle is presented in Appendix A at the end of the book. Required: 1. Calculate American Eagles percentage change in total assets and percentage change in net sales...
-
A compound with molecular formula C 9 H 10 O exhibits a strong signal at 1687 cm 1 in its IR spectrum. The 1 H and 13 C NMR spectra for this compound are shown below. Identify the structure of this...
-
A ketone with molecular formula C 9 H 18 O exhibits only one signal in its 1 H NMR spectrum. Provide a systematic (IUPAC) name for this compound.
-
In Poor Economics: A Radical Rethinking of the Way to Fight Global Poverty (Public Affairs Book 2012) by Abhijit Banerjee and Esther Duflo, read Chapter 2. How do you relate the story of Pak Solhin's...
-
(a) Based on the RSA algorithm, answer the following questions: i. Assume Alice has chosen 13 and 17 as two prime numbers (p and q), thus N=p.q=221. Define a value for e as her public key. Show your...
-
Explain the meaning of the following terms and how they are measured and comment on their use in the context of organizational management. a. Standard costing system b. balance scorecard approach c....
-
The first step in determining whether or not this is a worthy investment 6is to calculate the present value of the investment. In cell B10, calculate the present value of the investment from the...
-
1. For the following program (assume tempMeas global variable is updated by another thread): a) Construct the Control Flow Graph. b) The while loop in this program is non-terminating. Explain the...
-
X Pharmaceutical Ltd. looks for your assessment in regard of following bookkeeping exchanges: 1.Acquired a long term permit to make a specific medication at an expense of $1,00,00,000 toward the...
-
Given a linear program in slack form such that the basic solution is feasible, give an algorithm to find a vertex of the feasible region by optimizing the slack form.
-
Recall that Chapter 8 described the binary search algorithm for finding a particular entry in an ordered list. The idea behind binary search is to begin looking in the exact center of the list. If...
-
Rank the following alcohols in order of increasing ease of acid-catalyzed dehydration. OH OH OH
-
Acid-catalyzed dehydration of neopentyl alcohol, (CH3)3CCH2OH, yields 2-methyl-2- butene as the major product. Outline a mechanism showing all steps in its formation.
-
Acid-catalyzed dehydration of either 2-methyl-1-butanol or 3-methyl-1-butanol gives 2-methyl-2-butene as the major product. Write plausible mechanisms that explain these results.
-
Data governance provides Select one: A. Definitions, standards and procedures for how data is used. B. The internal data entry processes needed to capture accounting transactions. C. A road map that...
-
When it comes to cost allocation, there are various best practices that can be used. One is to thoroughly record the allocation process. This helps to guarantee that costs are allocated consistently...
-
Contractor Proposal Cost Calculation Table COST ELEMENT CHANGE ($) Material $5,000.00 Subcontracts $3,000.00 Direct Labor $2,000.00 Indirect Expenses (Fringe Only) (28.33%) $566.60 Other Direct...

Study smarter with the SolutionInn App


