All of the following compounds absorb IR radiation in the range between 1600 and 1850 cm -1
Question:
a.
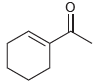
b.
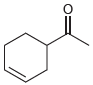
c.
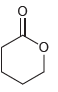
d.
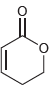
e.
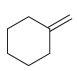
Transcribed Image Text:
O.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
a b c...View the full answer

Answered By

Gaurav Soni
Myself a student of Physics currently pursuing my PG. I helped many students due to my understanding specifically among students and also in my subject i. e. Physics. Helping them in various boards and in various entrance examination gives me a satisfaction of gaining knowledge. I have taught physics to students for many engineering and medical entrance examination and also for various boards in india. Till now have a great results from my students and that's why having a good impact on them. I have a good knowledge in conceptual and theoretical physics. Essential University Physics is one of my favorite field of teaching. I have a experience of almost 4 years in teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
All of the following compounds absorb infrared radiation between 1600 and In each case, 1. Show which bonds absorb in this region. 2. Predict the approximate absorption frequencies. 3. Predict which...
-
All of the following compounds can react as acids. Without using a table of acidities, rank them in order of increasing acidity. Explain your ranking. (a) CH3CH2SO3H (b) CH3CH2OH (c) CH3CH2COOH (d)...
-
What features will be similar in the IR spectra of the following compounds, and how will their IR spectra differ? H2C CHCHCH and CH CH CHCH,CHCH sDh
-
Find any horizontal or vertical asymptotes. f(x) = = X 1- X
-
The article by Gage-Bouchard discusses how social inequality manifests in health care settings, drawing from research on cultural health capital, fundamental cause theory and doctor-patient...
-
Parisian Cosmetics Company is planning a one-month campaign for September to promote sales of one of its two cosmetics products. A total of $140,000 has been budgeted for advertising, contests,...
-
Question: You are the CEO of a software company. You will only allow your engineers to create software for Apple computers, not for PCs because you think Apple is cooler and you hate Microsoft. Some...
-
OBrien Industries Inc. is a book publisher. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing balances are shown by letters. The...
-
pls solve asap. 1- Do you think that among the difficulties facing the application of the accrual basis of accounting is the lack of political will? 2- Does accounting on a cash basis cause...
-
Jaylen has asked you to make a retirement planning presentation to the employees at Troy Metals Inc. as part of a financial literacy day. Your presentation will last approximately 60 minutes with...
-
Does each of the following nucleophiles favor S N 2 or S N 1? a) b) c) (d) NaOH (e) NaCN SH
-
Rank each of the bonds identified in order of increasing wave number. R-CEN - N-H - -
-
Multiple sclerosis is a disease of the nervous system that causes severe weakness of the arms and legs, among many other damaging symptoms. Read the following excerpt from the abstract for this...
-
The electric field 69.0 cm from a very long uniform line of charge is 731 N/C. How much charge is contained in a 3.00 cm section of the line? Please give your answer in units of pC.
-
For each structure, draw a free-body diagram showing ONLY the non-zero reactions. You do not need to perform any calculations, but you will not get any points if you draw a reaction that, if...
-
Revenues $ 5 , 0 0 0 , 0 0 0 COGS $ 2 , 0 0 0 , 0 0 0 Gross Profits $ 3 , 0 0 0 , 0 0 0 Depreciation $ 5 0 0 , 0 0 0 EBIT $ 2 , 5 0 0 , 0 0 0 Interest $ 5 0 0 , 0 0 0 EBT $ 2 , 0 0 0 , 0 0 0 Taxes $...
-
The potential equals 7.92 V at the midpoint between two point charges that are 1.01 m apart. One of the charges is 1.06 x 10-9 C. Find the value of the other charge. Submit Answer
-
1. Select an actual service company related to or in tourism or hospitality (name of the hotel) and briefly describe what the organization does. Make sure to select an actual company . 2. Review the...
-
A quality control analyst randomly selects 3 different car ignition systems from a manufacturing process that has just produced 200 systems, including 5 that are defective. a. Does this selection...
-
White Bolder Investments (WBI) You are an intern working for WBI, a large investment advisory services in Sydney. Among other regular customers, WBI has been providing advisory services for Jumbo...
-
In each case, which group receives the higher priority? (a) (b) HC-CH (CH3)C
-
Which of the following two reactions should have the greatest ÎH° change? Why? (1) (2) C(CH3) (CH3)Q C(CH3) H C CH3 H3C CH3
-
Supply the curved-arrow notation for the acid-catalvzed isomerizatron.
-
On June 1 of the current year, Allen converted a machine from personal use to business use. At the time of the conversion, the machine was worth $40,000. Allen purchased the machine five years ago...
-
In chapter 19, what were risks involved in taking on the new Japanese customer contract? Question 27 options: A) The risk of currency changes which could result on eroding profits and causing losses...
-
Which feature is an indication of an efficient credit risk rating that meets regulatory requirements? Group of answer choices The system applies Machine Learning algorithmns. The rating discriminates...

Study smarter with the SolutionInn App


