Rank each of the bonds identified in order of increasing wave number. R-CEN - N-H - -
Question:
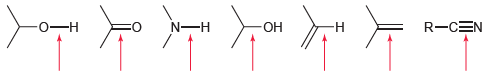
Transcribed Image Text:
R-CEN -о—н N-H -ОН -н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
OH NH In...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank each of the following series of compounds in order of increasing oxidationlevel: CI (a) (b) CH3CN CH3CH2NH2 H2NCH2CH2NH2
-
Rank the following bonds in order of increasing ionic character: NOO, CaOO, COF, BrOBr, KOF.
-
The partial Lewis structure that follows is for a hydrocarbon molecule. In the full Lewis structure, each carbon atom satisfies the octet rule, and there are no unshared electron pairs in the...
-
Write the expression as one ratio without any negative exponents. x1/4x-3/4 X
-
Discuss the growing income inequality in the U.S. and the world with someone mature enough to have some perspective from experience about it. Ask them what they think about the widening gap between...
-
La Femme Accessories Inc. produces women's handbags. The cost of producing 800 handbags is as follows: Direct materials............................................. $18,000 Direct...
-
Question: Uncle signed a lease with Landlord on a storefront in Georgetown, D.C. Uncle convinced Nephew to start a furniture store in the space. Uncle and Nephew formed a corporation to operate the...
-
Nayak Company has recorded the following items in its financial records. Cash in bank ............ $ 41,000 Cash in plant expansion fund ...... 100,000 Cash on hand ............ 8,000 Highly liquid...
-
Business Consulting. TASK 3 UPL's shareholders may or may not wish to participate in the buy-back depending on the potential tax impacts. This will depend on various factors, including the entity...
-
Japanese researchers have developed a compressiondepression method of testing electronic circuits based on Huffman coding (IEICE Transactions on Information & Systems, Jan. 2005). The new method is...
-
All of the following compounds absorb IR radiation in the range between 1600 and 1850 cm -1 . In each case, identify the specific bond(s) responsible for the absorption(s), and predict the...
-
How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy? Problem 15.29 a. b. OH HO. HO m/z = 126.0315 m/z = 126.1404
-
Pam Corporation has an 80 percent interest in Sun Corporation, its only subsidiary. The 80 percent interest was acquired on July 1, 2016, for $800, at which time Sun's equity consisted of $600...
-
For the group of stocks assigned to you, form the minimum variance frontier. What is the minimum attainable annualised standard deviation of the portfolio (using the 2014-2018 data pool): Q2: at an...
-
On 9 th November 2 0 2 1 , Ford and other five car manufacturers agree to sign COP 2 6 commitment, as part of efforts to cut carbon emissions and reduce global warming. You want to investigate...
-
Develop at least three planning metrics that represent different aspects of quality, are appropriate to the tools you chose, and are appropriate to your selected project. The metrics should reflect...
-
4 Part A The electric field at the point x = 5.00 cm and y = 0 points in the positive direction with a magnitude of 8.00 N/C. At the point a = 10.0 cm and y = 0 the electric field points in the...
-
5. (15 points) This question involves a simple example that illustrates why o can be estimated by when we compute the heteroskedasticity robust standard errors. Consider a simple regression model...
-
Adults were randomly selected for a Newsweek poll, and they were asked if they favor or oppose using federal tax dollars to fund medical research using stem cells obtained from human embryos. Of the...
-
Anna, a high school counselor, devised a program that integrates classroom learning with vocational training to help adolescents at risk for school dropouts stay in school and transition to work...
-
The industrial synthesis of methyl tert-butyl ether involves treatment of 2-methylpropene with methanol (CH3OH) in the presence of an acid catalyst, as shown in the following equation. CH3 H3C H3C...
-
The standard free energy of activation (G++) for hydration of 2-methylpropene to 2-methyl-2-propanol (Eq. 4.41, p. 169) is 91.3 kJ mol-t (Zt 8 kcal mol-l;. The standard free energy G for hydration of...
-
The standard free energy of activation (G++) for hydration of 2-methylpropene to 2-methyl-2-propanol (Eq. 4.41, p. 169) is 91.3 kJ mol-t (Zt 8 kcal mol-l;. The standard free energy G for hydration of...
-
Review the balance sheet for VWX Inc. as of December 31, 2023, to understand its financial stability and liquidity. VWX Inc. Balance Sheet As of December 31, 2023 Assets Current Assets Cash $30,000...
-
U.S. Generally Accepted Accounting Principles (US GAAP) and the American Institute of Certified Public Accountants (AICPA) are often considered a champion for principles-based standards. Group of...
-
Alex provides $20,000 of legal services in exchange for a 40% capital and profits interest in B partnership. What income or deductions must be picked up in Alex's and B Partnerhip's tax returns?...

Study smarter with the SolutionInn App


