Assign IUPAC names for each of the following compounds: (a) (b) (c) (d) . CH
Question:
(a)
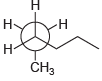
(b)
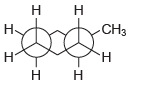
(c)
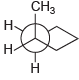
(d)
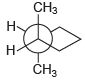
Transcribed Image Text:
Н Н н н. CH н н На CH3 Н н н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a Hexane b Meth...View the full answer

Answered By

Moses mwangi
With prior writing experience, be sure that I will give a great grade, If not an A+, it will be something close to this. My reviews speaks it all, Try me!!
4.80+
78+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Assign an IUPAC name for each of the following compounds. a. b. c. d. e. f. g. SH
-
Assign an IUPAC name for each of the following compounds: a. b. c. d. ,
-
Give the IUPAC names for each of the following compounds: (a) CH2CH(CH2)5CHCH2 (b) (c) (CH2CH)3CH (d) (e) (f) CH2CCHCHCHCH3 (g) (h) CH3 CHz CH3 HH CI CI H H H3C CH,CH2 CH2CH3
-
Table # 11.1.1: Autism Versus Breastfeeding Breast Feeding Timelines Autism Yes No None 241 20 Column Total 261 Less than 2 months 198 25 223 2 to 6 months 164 27 191 More than 6 months 215 44 259...
-
Design an ergonomic handle indicating all of the principles used in the design.
-
How would you prepare 2.00 L of (a) 0.10 M KOH from the solid? (b) 0.010 M Ba(OH)2 8H2O from the solid? (c) 0.150 M HCl from a reagent that has a density of 1.0579 g/mL and is 11.50% HCl (w/w)?
-
The managing partner of Singh Accounting is considering the desirability of tracing more costs to jobs than just direct labour. In this way, the firm will be better able to justify billings to...
-
Austin Landers is the accountant for Sun Coast, a manufacturer of outdoor furniture that is sold through specialty stores and internet companies. Annually, Landers is responsible for reviewing the...
-
During 2017, Blevert Co introduced a new line of machines that carry a three-year warranty against manufacturer's defects. Based on industry experience warranty couts are muted at 2% of sales in the...
-
The results of a time study to perform a quality control test are shown in the following table. On the basis of these observations, determine the normal and standard time for the test, assuming a 23%...
-
Identify the product(s) in each of the following reactions: (a) (b) (c) (d) Br ETOH
-
Some plants grow in soils as high as 20% iron. If the iron is present in the form of Fe(OH)3, the plants can still be iron deficient. Explain how this can be true. How can the iron in the soil be...
-
On January 1, 2011, Delphin Ltd. acquired all the share capital of Telescon Ltd. when Telescons equity consisted of: 100,000 common shares issued at $0.75 each... $75,000 Retained earnings...
-
1. 7.310 km 5.70 km = 2. (7.8m 0.34m) (1.15s + 0.82s) =
-
In the year 2005, a picture supposedly painted by a famous artist some time after 1715 but before 1765 contains 99.8 percent of its carbon-14 (half-life 5730 years). From this information, could this...
-
During 2021, Peoria Airlines paid salary expense of $40.8 million. At December 31, 2021, Peoria accrued salary expense of $2.3 million. Peoria then paid $2.6 million to its employees on January 3,...
-
The following T-account is a summary of the Cash account of Swifty Company. Cash (Summary Form) Balance, Jan. 1 Receipts from customers 8,200 362,100 Payments for goods 294,200 Dividends on stock...
-
A spherical, non-conducting shell of inner radius 1= 10 cm and outer radius 12= 15 cm carries a total charge Q = 15 C distributed uniformly throughout the volume of the shell. What is the magnitude...
-
Consider a drill press containing three drill bits. The current policy (called individual replacement) is to replace a drill bit when it fails. The firm is considering changing to a block replacement...
-
Using Apple, demonstrate how the differentiation strategy can be well implemented.
-
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is...
-
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is...
-
One of the isomers of C 5 H 12 reacts with Cl 2 in the presence of light to produce three isomers of C 5 H 11 Cl: This reaction replaces am one of the hydrogen?s of C 5 H 12 with a Cl. What arc the...
-
What is a good way to improve efficiency and accuracy in the registration process? a. Email audit results to staff members with instructions to improve. Training takes up too much time. b. Scan a...
-
Should Burton pursue both the thermowell machine purchase and the EE acquisition? Why or why not
-
The work sheet: multiple choice eliminates any need for an audit of the financial statements. is often made available to external decision makers. aids in the preparation of financial statements....

Study smarter with the SolutionInn App


