Identify the product(s) in each of the following reactions: (a) (b) (c) (d) Br ETOH
Question:
Identify the product(s) in each of the following reactions:
(a)
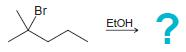
(b)
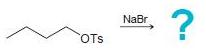
(c)
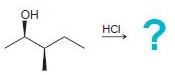
(d)
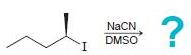
Transcribed Image Text:
Br ETOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a b...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the product of each of the following reactions: a. b. c. d. CH CH3 A CH2CH3 CH2CH 2CH3 -A CH2CH3 h CH2CH3 CH2CH3 CH2CHs
-
Predict the product(s) in each of the following aldol condensations. acetophenone + hexanal + hexanal NH
-
Identify the alkene product in each of the following Wittig reactions: (a) (b) ButanaH5)2P CHCH CH2 Cyclohexyl methyl ketone(H)2P CH2
-
Which statement is correct? A) Tax credits reduce tax liability on a dollar-for-dollarbasis. B) Tax deductions reduce tax liability on a dollar-for-dollarbasis. C) The benefit of a tax credit depends...
-
What are the key concerns in the design of a power tool?
-
A series of solutions containing NaOH, Na2CO3, and NaHCO3, alone or in compatible combination, was titrated with 0.1202 M HCl. Tabulated below are the volumes of acid needed to titrate 25.00-mL...
-
You are asked to bring the following incomplete accounts of a printing plant acquired in a merger up to date through January 31, 2006. Also consider the data that appear following the T-accounts....
-
Global Green Books Publishing produces customized eBooks for a local college. It has just received an order for a new eBook on Strategic Human Resource Management in a Global Context from a senior...
-
Information for two alternative projects involving machinery investments follow. The accounting rate of return for Project 1 is: Project 1 Year Project 2 Initial investment $ (330,000) $ (270,000)...
-
Is it true, as Jack Carter claims, that virtually all of our workers are women or minorities anyway, so no one can come in here and accuse us of being discriminatory?
-
Draw a relative energy diagram showing the conformational analysis of 1,2-dichloroethane. Clearly label all staggered conformations and all eclipsed conformations with the corresponding Newman...
-
Assign IUPAC names for each of the following compounds: (a) (b) (c) (d) . CH CH3
-
British Airways, Plc. (BA), a U.K. company, prepares its financial statements according to International Financial Reporting Standards. BA's annual report for the year ended March 31, 2009, which...
-
If f'(x) is continuous, f(-5) = 19, and f(-8)= 21 x L -8 f'(x) dx = -2, find f(-8). -5 Recall the second part of the Fundamental Theorem of Calculus. If g is a continuous function on the interval [a,...
-
1. Calculate this integral. Show and annotate the various steps in the process. (6) x +3 da 49
-
Find two positive numbers with product 484 and whose sum is a minimum. Enter your answers in increasing order. First number: Number Second Number: Number
-
Find dy/dx for the function. dy dx 11 x 10y - 8 = 0
-
[6 points] For the given integral x 4 - x a. Evaluate the integral using trig substitution. b. Evaluate the integral using u-substitution. dx
-
Mary Higgins is a freelance writer with enough spare time on her hands to play the stock market fairly seriously. Each morning she observes the change in stock price of a particular stock and decides...
-
For the given transfer function: Vo(s) / Vi(s) = (s^2C^2R^2 + 1) / (s^2C^2R^2 + 4sCR + 1) Assumiing that 1/(CR) = 120 PI so write the matlab code to find the magnitude plot
-
Benzene and hexane are both liquids at room temperature. Do you expect benzene and hexane to be miscible? Do you expect benzene and water to be miscible? Explain. Hexane 0 Benzene
-
One of these isomers is miscible with water, and the other is nearly insoluble. Explain. CHCHCHCOH CH,COCHCH3
-
Because of two hydrogen bonds, carboxylic acids show a very strong attractive force between two molecules that persists even in the gas phase. Show this hydrogen bonding between two carboxylic acid...
-
s CB1(1) and YA1 of the Income Tax Act 2007 are very important because: Question 5Answer a. these sections explain how an employee, sole trader, partnership, trust and company should be treated...
-
If the seller is responsible for the shipping costs of merchandise sold, the shipping terms will be specified as:Multiple choice question.FOB destinationFOB factoryFOB shipping pointFOB start
-
A golf club production company wants to ensure that all aspects of its production and distribution processes are operating at optimal efficiency. The company produces three types of clubs, a line for...

Study smarter with the SolutionInn App


