For each of the following compounds determine which (if any) lone pairs are participating in aromaticity. a.
Question:
a.
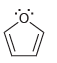
b.
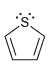
c.
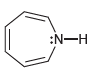
d.
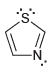
e.
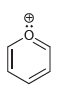
f.
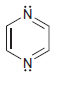
g.
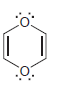
h.
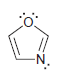
Transcribed Image Text:
:N-H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a One of the lone pairs on oxygen b One of the lone pairs on s...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, determine whether or not you would expect its IR spectrum to exhibit a signal to the left of 3000 cm -1 a. b. c. . d. e. f.
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) OMe . CI H,
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
State Newtons second law of motion. What are the limitations on the use of Newtons second law? Explain.
-
The New England Cycle Company is planning next month's production of its two bicycles: its B2 tandem cycle built for two and its S10 single-rider 10-speed model. Both models use the same seats and...
-
Determinants of Sums Give an example of square matrices A and B for which |A + B| ( |A| + |B|?
-
What is the cost of capital equal to?
-
J&J Snack Foods Corp. is a national manufacturer and distributor of snack foods and frozen beverages. Among its brands are ICEE, SuperPretzel, and Cinnabon CinnaPretzel. Adapted versions of the...
-
Problem 10 The Verry Place (VP), open 365 days a year, consists of a 100-room hotel with a 50-seat restaurant shop. Sandy Verry provides you with the following information: 1. Of the 100 rooms, 70...
-
Go to the Webinars worksheet. DeShawn wants to determine the number of webinars the company can hold on Tuesdays and Thursdays to make the highest weekly profit without interfering with...
-
Identify which of the following compounds is more acidic and explain your choice.
-
Go to the beginning of Section 18.1 where 8 best-selling drugs were shown. Review the structures of those compounds, and identify all of the aromatic rings that are not already highlighted in red. ...
-
Use the linspace function to create vectors identical to the following created with colon notation: (a) t = 4:6:35 (b) x = -4:2
-
What may be five training topics that human relations representatives should include in training sessions for the supervising employees in an organization? Why would the five areas of your choice be...
-
Leaders can inadvertently demotivate employees by what? 1 point setting goals in collaboration with employees micromanaging and hand-holding employees encouraging employees to think independently...
-
4-20. The equation for a Gaussian curve is 1 -(x-1)/20 V2TT In Excel, the square-root function is SQRT and the exponential function is EXP. To find e 3.4, write EXP(-3.4). The Gaussian exponential...
-
Participants in their study first completed two tasks, a placekeeping task [UNRAVEL] and a Psychomotor Vigilance Task [PVT] and then were randomly assigned to either stay up all night in the lab, or...
-
Your managers and workers require copies of the adjusted plan. Some of your workers do not have access to electronic systems. How will you supply this to them?
-
Describe how to compute shiftHash(h(X[i..i+m1]), X, i) for the hash function, h(X[i..i + m 1]) = X[i] + + X[i + m 1], where each character is viewed as an integer in the range [0, c 1], with c...
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
Would you expect the reaction of propyl bromide with sodium cyanide (NaCN), that is, CH3CH2CH2Br + NaCN CH3CH2CH2CN + NaBr to occur faster in DMF or in ethanol? Explain your answer.
-
When tert-butyl bromide undergoes solvolysis in a mixture of methanol and water, the rate of solvolysis (measured by the rate at which bromide ions form in the mixture) increases when the percentage...
-
List the following compounds in order of decreasing reactivity toward CH3O- in an SN2 reaction carried out in CH3OH: CH3F, CH3Cl, CH3Br, CH3I, CH3OSO2CF3, 14CH3OH.
-
q1: q2: help and explain The Museum of History is preparing for its annual appreciation dinner for contributing members. Last year, 650 members attended the dinner. Tickets for the dinner were $26...
-
This is part 2. The following information is from the Oregon Revised Statutes, Chapter 673, for Tax Consultants and Tax Preparers. Click the following link to access the reading material for part 2...
-
The manager explained that this practice created a pool of money that could be refunded to departments when they were having financial difficulties. It's also known that department managers receive a...

Study smarter with the SolutionInn App


