Predict the chemical shifts for the signals in the 1 H NMR spectrum of each of the
Question:
(a)
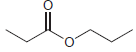
(b)
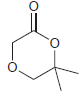
(c)
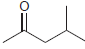
(d)
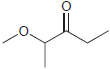
(e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a b c d e All four methylene groups are equivalent so the compound will have only one signal in ...View the full answer

Answered By

Zablon Gicharu
I am an educator who possesses the requisite skills and knowledge due to interacting with students for an extended period. I provide solutions to various problems in step-by-step explanations, a well-thought approach and an understandable breakdown. My goal is to impart more straightforward methodologies and understanding to students for more remarkable achievements.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the chemical shifts of the protons in the following compounds. (a) (b) (c) (d) (e) (f) HC(CH3)3 CH CH CH3 , - CH,CCCH OH CH,CH,-C-OH CH3 CHC-CH Br
-
Predict the approximate chemical shifts for the different hydrogen's in thesecompounds: CI CI a) CH,CH,CH3 b) CH;CHCH3 c) CH,COCH,CH3 d) CH;CHCH2
-
Explain the direction of the chemical shifts for Fe(0), Fe(II), and Fe(III) in Figure 22.20. Fe 2p at normal emission Fe 2p32 Fe 2p12 Fe (0) Fe/MgO(001) Fe(0) Fe;OM9O(001) Fe(ll & II) Fe(ll) Fe(lI)...
-
Net sales revenue, net income, and common stockholders' equity for Vallen Optical Corporation, a manufacturer of contact lenses, follow for a four-year period. Requirements 1. Compute trend...
-
What is a "liquidity premium?" When do investors increase their attention to such a premium?
-
Solve diagrams (a)-(c) for the unknowns R, S, and T, assuming a 10% interest rate. (a) (b) (c) 100 100 100 00 150 100 50 30 60 0120 0234-5
-
A sales invoice prepared by the seller is called a purchase invoice by the buyer. True/False
-
Cheryl Montoya picked up the phone and called her boss, Wes Chan, the vice president of marketing at Piedmont Fasteners Corporation: Wes, Im not sure how to go about answering the questions that came...
-
What is the present value of a security which pays the certain cash flow streams of $100 at the end of the first year, $80 at the end of the second year, $90 at the end of the third year, and $130 at...
-
Although the ultimate goal of the parties in a contract is to discharge the obligations of the parties by performing as promised, a contract may also be discharged through substantial performance,...
-
Identify the structure of a compound with molecular formula C 9 H 20 that exhibits four CH 2 groups, all of which are chemically equivalent. How many total signals would you expect in the 1 H NMR...
-
A 1 H NMR spectrum was acquired for each of the following constitutional isomers. Comparison of the spectra reveals that only one of these spectra exhibits a signal between 6 and 7 ppm. Identify the...
-
Despite Operational Excellence providing the perfect platform to push Risk Management, discuss what the potential pitfalls may be.
-
What is the difference between technology sourcing and technology procurement?
-
Which accounting enterprise architecture management competency is likely to have the greatest positive impact on an accountants career?
-
Which accounting role in enterprise architecture management is the most critical to building and maintaining that capability? Why?
-
What is the difference between privacy and information privacy?
-
What is the difference between cybersecurity management and cybersecurity?
-
In Oracle, what is a tablespace?
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
Name the five isomers of C6H14. Discuss.
-
Explain why each of the following names is incorrect: (a) 2, 2-Dirnethyl-6-ethytheptane (b) 4-Ethyl-5, 5-dirnethylpentane (c) 3.Ethyl-4, 4-dimcthylhexane (d) 5, 5, 6-Trimcthyloctane (e)...
-
Propose structures and give IUPAC names for the following: (a) A diethyldimethyihexane (b) A (3-methylbutyl)-substituted alkane
-
GNI per capita is not always the best measure of actual total annual income because in some countries, many business transactions take the form of: Group of answer choices treasury yields tax...
-
To determine cash flows from investing activities, an accountant will usually start with the ledger balance of assets and liabilities (that are not operating) and investigate the individual...
-
Operating statements include all of the following except Group of answer choices Asset values Depreciation Personnel expenses Revenues

Study smarter with the SolutionInn App


