Predict the major product(s) for each of the following reactions: 1) MCPBA 1) , THF 2),, NaOH
Question:
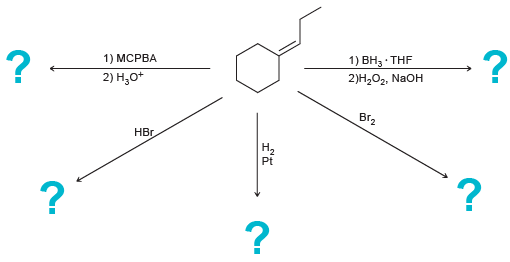
Transcribed Image Text:
1) MCPBA 1) ВН, THF 2)Н,О, NaOH 2) Н,о* ВГz НЕ. На Pt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
HO OH En Br 1 M...View the full answer

Answered By

Hamza Amjad
Currently I am student in master degree program.from last two year I am tutring in Academy and I tought many O/A level student in home tution.
4.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
Assume that 3-month Treasury bills totaling $23 billion were sold in $10,000 denominations at a discount rate of 5.200%. In addition, the Treasury Department sold 6-month bills totaling $21 billion...
-
Develop and prototype a new interface design for the system's function using Graphical User Interface. Include common interface functions such as (textboxes, radio button, drop-down menus, check...
-
In Exercise 1 - 2, either use an appropriate theorem to show that the given set, W, is a vector space, or find specific example to the contrary? 1. 2. b: a +b+c= 2 Lc.
-
For each of the definitions, unite the letter of the appropriate term in the space provided. 1_The revenue account used to record a sale. 2-The revenue account used to record a sales return. 3-Window...
-
A cylinder of radius ro, length L, and thermal conductivity k is immersed in a fluid of convection coefficient hand unknown temperature T. At a certain instant the temperature distribution in the...
-
Question 4 - After aging the accounts receivable, it is estimated that $700 will not be collected and the allowance account has an existing credit balance of $100. The adjusting entry under the aging...
-
Skylar and Walter Black have been married for 25 years. They live at 883 Scrub Brush Street, Apt. 52B, Las Vegas, NV 89125. Skylar is a stay-at-home parent and Walt is a high school teacher. His W-2...
-
Would water be a suitable proton source to protonate the following compound? ONa
-
When 3-bromo-3-ethylpentane is treated with sodium acetylide, the major products are 3-ethyl-2-pentene and acetylene. Explain why the carbon skeleton does not change in this case, and justify the...
-
If possible, solve the system. =6 x + 3y + z = 6 + + 3x + y - z = 6 x-y-z= 0 =
-
In a given month, a restaurant has an opening inventory of $2,700 and a closing inventory of $2,000. If the cost of food sold for that month is $5,100, what is the inventory turnover rate?
-
A building is expected to generate no cash flows for several years and then generate annual cash flows forever.What is the value of the building if the first annual cash flow is expected in 7 years,...
-
Based on CIA (Covered Interest Arbitrage), the spot Yen/US$ exchange rate is Yen100/US$ (i.e., 100 Japanese yen per 1 U.S. dollar) and the one year forward rate is Yen200/US$. If the annual interest...
-
At the beginning of this year, a firm had $30M in cash, $20M in accounts receivable, $45M in inventory and $10M in taxes payable. Going forward, the firm plans to finance some of its inventory with...
-
You are considering a project that will cost you $60,000 today and is projected to earn $5,000 next year, with the inflows growing at a 4% annual rate in perpetuity thereafter. If your cost of...
-
A retailer is considering two possible definitions of in stock: I. A product is in stock if it has at least one unit on hand at the end of the day. II. A product is in stock if it has satisfied all...
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
Explain why the reaction of the cis-isomer of this compound with potassium tert-but oxide in tert-butanol is about 500 times faster than that of thetrans-isomer. Br C(CH3)3
-
Explain which of these compounds has a faster rate of E2elimination: CH CH3 CI 'CI
-
Frequently, several different routes can potentially be used to synthesize a desired compound. For example, the following two routes can be envisioned for the preparation of cyclopentyl methyl ether....
-
Reimel Furniture Company, Inc. incurred the following costs during 20x2. Direct material used Direct labor Manufacturing overhead $174,000 319,000 160,000 During 20x2, products costing $121,000 were...
-
Helix Corporation uses the weighted-average method in its process costing system. It produces prefabricated flooring in a series of steps carried out in production departments. All of the material...
-
Groucho, Harpo, and Chico form a partnership on January 1, 2021. Groucho contributes $270,000, Harpo $210,000, and Chico $120,000 to a business called Marx Brothers' Partnership. On a monthly basis,...

Study smarter with the SolutionInn App


