Would water be a suitable proton source to protonate the following compound? ONa
Question:
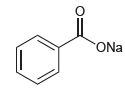
Transcribed Image Text:
ONa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
No water would not be a suitable proto...View the full answer

Answered By

HABIBULLAH HABIBULLAH
I have been tutor on chegg for approx 5 months and had solved a lot of questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
We will learn all of the following reactions in upcoming chapters. For each of these reactions, notice that the product is an anion (ignore the positively charged ion in each case). In order to...
-
Would ethanol (CH 3 CH 2 OH) be a suitable solvent in which to perform the following proton transfer? Explain your answer: ONH2 + NH3 NH2 H.
-
The following compound is known to be chiral. Draw its enantiomer, and explain the source of chirality. CH
-
Prepare a company analysis (strengths and weaknesses) Instructions - Prepare the SW portion of a SWOT analysis as a report. Use your textbook and the instruction below. Prioritizing the Results: The...
-
Investigate learning management software packages that are available through application service providers. Using the World Wide Web, identify at least two potential sources of such software. What...
-
Prove the orthogonality relations (28.4) for the trigonometric system. Equation 28.4 (-T,T) cos(jx) cos(kx) dx = (-,) L sin (lx) sin(mx) dx = (-TT) 0, , 2, if lm, T, if l=m, cos(kx) sin(x) dx=0 for...
-
Jill Riggins owns and operates Jills Java coffee shop. She proposes to account for the shops assets at their current market value in order to have current amounts on the balance sheet. Which...
-
Presented below are the consolidated work paper balances of Bush, Inc., and its subsidiary, Dorr Corporation, as of December 31, 2016 and 2015: Additional information: a. On January 20, 2016, Bush,...
-
Given f(x) = x-5x, (a) Find f(x+h) and simplify. f(x+h)-f(x) (b) Find and simplify. h Part: 0 / 2 Part 1 of 2 (a) f(x+h) = G
-
The subsidiaries of Perth International remit their earnings and investment proceeds to the Australian parent at the end of each year. The annual weighted average cost of capital or required rate of...
-
There are four constitutional isomers with molecular formula C 3 H 9 N. Draw a Lewis structure for each isomer and determine the number of lone pairs on the nitrogen atom in each case?
-
Predict the major product(s) for each of the following reactions: 1) MCPBA 1) , THF 2),, NaOH 2) ,* z . Pt
-
The frequency of the highest note on the piano is 4,186 Hz. (a) How many harmonics of that note can we hear? (b) How many harmonics of the note one octave below it can we hear?
-
In 2019, Maggy (34 years old) is an employee of YBU Corp. YBU provides a 401(k) plan for all its employees. According to the terms of the plan, YBU contributes 50 cents for every dollar the employee...
-
Which capital budget technique always yields the correct result and why?
-
Matthew (48 at year-end) develops cutting-edge technology for SV Inc., located in Silicon Valley. In 2019, Matthew participates in SVs money purchase pension plan (a defined contribution plan) and in...
-
On their weekly visits to a local restaurant, Bram and Mila usually buy one slice of appeltaart (Dutch apple pie) to share and two cups of coffee each. However, the restaurant offers a special...
-
John (age 51 and single) has earned income of $3,000. He has $30,000 of unearned (capital gain) income. a. If he does not participate in an employer sponsored plan, what is the maximum deductible IRA...
-
In 2018, Meg Inc, discovered a n error in its 2015 financial statements. The firm recorded $11,000,000 of depreciation expense on its equipment instead of recording $10,000,000. Meg has a constant...
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
Show the products of this reaction. How would the composition of the products change if t-BuO ? in t-BuOH were used in place of ethoxide ion in ethanol? ELOH + CH,CH,0
-
All of the stereo isomers of 1, 2, 3, 4, 5, 6-hexachlorocyclohexane have very similar rates of E2 reaction except the following stereo isomer, which reacts about 7000 times more slowly than the...
-
Show the product of thisreaction: Ph Br- - + NaOEt Br EIOH Ph
-
Role play Accounts and ledgers - debtor management the student to participate and facilitate a role play meeting with Peter Crowe, a client from Hurst & Smith Plumbing Pty Ltd, who owes Azure Beach...
-
Conduct a financial analysis of the firm including calculating ratios in the following areas: Liquidity Asset management Debt Management Profitability Internal and Sustainable Growth Write Executive...
-
On June 8, Sheridan Ltd. was incorporated and issued 34,200 common shares for $342,000. On August 19, an additional 8,550 shares were issued for $102,600. On November 2, the company paid $32,832 to...

Study smarter with the SolutionInn App


