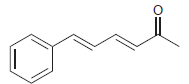
Use resonance structures to help you identify all sites of low electron density (δ+) in the following
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
5...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use resonance structures to help you identify all sites of high electron density (δ-) in the following compound:
-
Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H bond. Draw the structures of the anions resulting from loss of H+ from phenol and methanol, and use...
-
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain why none of the other regioisomer isformed. HCI CI CI
-
Consider the cash flows in Table P6.7 for the following investment projects (MARR = 15%). Determine the annual equivalent worth for each project at i = 15% and determine the acceptability of each...
-
Video Concepts, Inc. (VCI) manufactures a line of DVD recorders (DVDs) that are distributed to large retailers. The line consists of three models of DVDs. The following data are available regarding...
-
Rewrite the program of Table 4.1 to eliminate all array variables.
-
Subject to what conditions is it possible to compare EPS before and after a deal?
-
(a) Explain the similarities and differences between standards and budgets. (b) Contrast the accounting for standards and budgets.
-
Exercise 20-6 (Algo) Manufacturing: production budgets LO P1 Electro Company manufactures transmissions for electric cars. Management reports ending finished goods inventory for the first quarter at...
-
If Wild Widgets, Inc., were an all-equity company, it would have a beta of .85. The company has a target debtequity ratio of .40. The expected return on the market portfolio is 11 percent, and...
-
Suppose that you measured the product PV of 1 mol of a dilute gas and found that PV = 24.35 L atm at 0.00C and 33.54 L atm at 100.C. Assume that the ideal gas law is valid, with T = t(C) + a, and...
-
Liquid N 2 has a density of 875.4 kg m 3 at its normal boiling point. What volume does a balloon occupy at 298 K and a pressure of 1.00 atm if 3.10 10 3 L of liquid N 2 is injected into it? Assume...
-
Find a linear differential operator that annihilates the given function. e -x + 2xe x - x 2 e x
-
Compensation data for numerous jobs are more available due to the existence of various websites. The website of Salary.com provides pay data for numerous jobs in different companies in different...
-
Imagine that you are the supervisor of the transcription group of the health information services department at Community Hospital. Your crew consists of several transcriptionists who handle, in...
-
1. A soccer player is on the edge of the field 30m from the goal line. To get it past the goalie, the player needs to direct the ball so that it is 15m in from the edge of the field. At what angle...
-
An equipment acquisition proposal is being considered by a large healthcare organization, XYZ Health Care. The array machine will enable the hospital to perform autoimmunity tests (for...
-
Using the case for leadership entitled "Surprise at St. Clair Middle School. Answer the questions below. 1. Do you have a problem here? If so, what is it? 2. How should you share the data with your...
-
Describe an O(n log n)-time algorithm for finding the second closest pair of points in a set, S, of n points in the plane. That is, you should return the pair, (p, q), in S, such that the only pair...
-
Would you use the adjacency matrix structure or the adjacency list structure in each of the following cases? Justify your choice. a. The graph has 10,000 vertices and 20,000 edges, and it is...
-
Synthesize the following compound starting with ethyne and 1-bromopentane as your only organic reagents (except for solvents) and using any needed inorganic compounds. Br Br
-
Predict features of their IR spectra that you could use to distinguish between the members of the following pairs of compounds. You may find the IR chart in the endpapers of the book and Table 2.1...
-
Deduce the structures of compounds A, B, and C, which all have the formula C6H10. As you read the information that follows, draw reaction flowcharts (roadmaps) like those in Problems 8.24 and 8.52....
-
It has been said that the next generation of auditing seeks to take full advantage of emerging technologies in a way that will improve audit quality. Which emerging technology do you believe has the...
-
Which of the following options is not considered a qualified adoption expense under the IRC? A. Expenses incurred that are not reimbursed under another program. B. Expenses directly related to the...
-
Define the Salaries payable account by selecting the appropriate statement below. Multiple choice question. It reports amounts owed to employees and is a liability. It reports amounts owed to...

Study smarter with the SolutionInn App


