Using the data in the following table, predict the sign and magnitude of ÎH° for each of
Question:
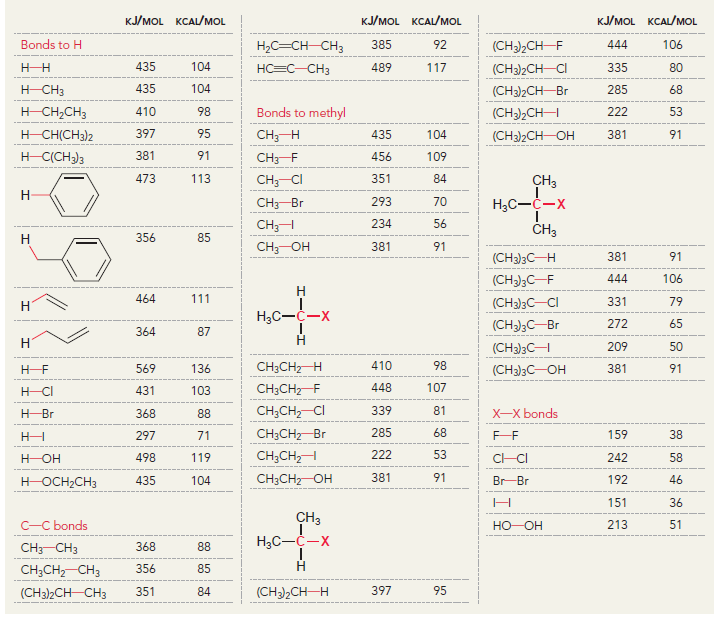
a.
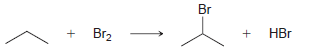
b.

c.
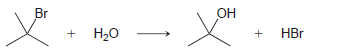
d.

Transcribed Image Text:
KJ/MOL KCAL/MOL к./мOL KCAL/MOL кJ/мOL KCAL/мOL Нас—сн сHз (CH3)2CH F (CH3)2CH CI Bonds to H 385 92 444 106 435 104 НC-С-СНз 489 117 335 80 H CH3 435 104 (CH3)2CH-Br 285 68 н сн-CHз Bonds to methyl 410 98 222 53 (CH3),CHH н СНICH)2 397 95 CHH 435 104 (CH3)2CH-OH 381 91 н ССH)з 381 91 456 109 CH3F CH3 473 113 351 84 CH; CI Н Нас—С—х 293 70 CH3-Br 234 56 CH;H CHз 356 85 Н Cн ОН 381 91 (CH)3C— Н 381 91 (CH)3C-F 444 106 Н 111 464 (CH3)3C-CI 331 79 Н Нас—с—х 272 65 (CHС—Вг 364 87 Н Н (CHд)3C— 209 50 CH3CH2 H 410 98 569 136 (CH)3с—ОН 381 91 448 107 CH3CH2 F H-CI 431 103 CH;CH,-CI 339 81 X-X bonds HBr 368 88 CH3CH2 Br 285 68 159 38 нн 297 71 222 53 CH;CH,H 498 119 C-CI 242 58 H-OH Cн CHz—ОН 381 91 435 192 46 HOCH2CH3 104 Br Br 151 36 CHз C-C bonds 213 51 но-ОН HаС —с —х CH CНз 368 88 Н CH,CH CHЗ 356 85 (CH)2CH H 351 397 95 84 (CH3)2CH CH3 Br Br, НBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a Bonds Broken kJmol Bonds Formed kJmol H x CH CH 3 2 397 CH 3 2 CH x Br 285 Br x Br 192 H x Br 368 ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the data in the following table, calculate the pI of the following amino acids. (a) Aspartic acid (b) Leucine (c) Lysine (d) Proline THE PK,VALUES FOR TWENTY NATURALLY OCCURRING AMINO ACIDS...
-
Using the data in the following table, calculate the pI of the following amino acids: (a) l-Alanine (b) l-Asparagine (c) l-Histidine (d) l-Glutamic acid THE PK,VALUES FOR TWENTY NATURALLY OCCURRING...
-
Carbon disulfide (CS2) is a toxic, highly flammable substance. The following thermodynamic data are available for CS2(l) and CS2(g) at 298 K: (a) Draw the Lewis structure of the molecule. What do you...
-
Construct the general solution of x ' = Ax involving complex eigenfunctions and then obtain the general real solution. Describe the shapes of typical trajectories. =[ A = 3. -2 1]
-
Provide at least two examples of police corruption as it relates to drugs. Discuss some of the strategies that agencies are implementing to combat corruption in the United States or other countries.
-
Where does this matrix send the standard basis for R2? Any other bases? cos(20) sin(20) sin(20) cos(20)
-
What is the natural temptation of a company that is required to pay out 100% of its earnings, in terms of how much earnings it records?
-
Barkley Company's adjusted trial balance on March 31, 2015, its fiscal year-end, follows. On March 31, 2014, merchandise inventory was $37,500. Supplementary records of merchandising activities for...
-
27-28 Find the exact area of the surface obtained by rotating the given curve about the x-axis. 27 v = 1/r
-
Pane records depreciation and amortization expense. Use the nearest whole month convention, and the double declining balance method for depreciation and the whole-year convention and the...
-
As mentioned in problem 5.58, some molecules are chiral even though they lack a chirality center. For example, consider the following two compounds shown, and explain the source of chirality in each...
-
For each of the following processes predict the sign of ÎS for the reaction. In other words, will ÎS sys be positive (an increase in entropy) or negative (a decrease in entropy)? a. b. c....
-
The Happy Times, Limited, company of Arizona has budgeted sales of its most popular alphabet game for children ages 4 through 8 years. The budget is for the next four months, beginning in April....
-
On 3/1/18 Comprehensive Corp. issues $100,000, 8-year bonds (dated 3/1/18) with a stated rate of 3% payable semiannually every 9/1 and 3/1. The proceeds of the bonds are $93,211. Comprehensive Corp....
-
6) Use exponential smoothing with trend adjustment to forecast deliveries for period 10. Let alpha = 0.4, beta = 0.2, and let the initial trend value be 4 and the initial forecast be 200. Actual...
-
4) ABC Corporation makes electrical watches. The lifetime (period during which a watch does not need major repair) of an electronic watch manufactured by this corporation has a normal distribution...
-
Russel Industries has just completed construction of an oil drilling facility at a cost of $10 million. The facility has a useful life of 10 years, is expected to have a $1 million residual value,...
-
write java programme Algorithm: Array Average (Array, n) Purpose: Find the average value Pre: Given the array Array and the number of elements, n Post: None Return: The average value { } } sum -0.0...
-
Give an alternative pseudocode description of Kruskals algorithm that makes explicit use of the union and find operations.
-
Diamond Walker sells homemade knit scarves for $25 each at local craft shows. Her contribution margin ratio is 60%. Currently, the craft show entrance fees cost Diamond $1,500 per year. The craft...
-
For all practical purposes, the compound cyclohexa-2, 4-dien-1-one exists totally in its enol form. Write the structure of cyclohexa-2, 4-dien-1-one and of its enol form. What special factor accounts...
-
How would you use the acetoacetic ester synthesis to prepare the following?
-
How would you use the acetoacetic ester synthesis to prepare the following?
-
Villian, Inc.'s comparative income statement follows. The 2023 data are given as needed.Begin by selecting the formula to calculate the profit margin ratio.Profit margin ratioXData tableTeal'S Clued...
-
When completing a bank reconciliation, Question 1 options: a) enter the check register balance on the current date. b) add bank fees to the check register amount. c) subtract outstanding deposits...
-
2. PT. Asindo pada tahun 2021 memiliki informasi sebagai berikut. Saldo kas awal Rp 10.000.000 Laba yang ditahan, awal Rp 15.000.000 Persediaan awal 5.000 Unit @Rp 3.500 Rp 17.500.000 Pembelian 7.000...

Study smarter with the SolutionInn App


