As mentioned in problem 5.58, some molecules are chiral even though they lack a chirality center. For
Question:
a.
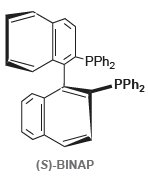
b.
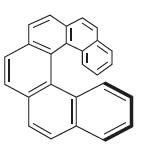
Transcribed Image Text:
PPH2 PPH2 (S)-BINAP
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a This compound cannot be completely planar because steric hindrance prevents the two ri...View the full answer

Answered By

Robert Mwendwa Nzinga
I am a professional accountant with diverse skills in different fields. I am a great academic writer and article writer. I also possess skills in website development and app development. I have over the years amassed skills in project writing, business planning, human resource administration and tutoring in all business related courses.
4.90+
187+ Reviews
378+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1 H NMR spectroscopy? c) 13 C NMR spectroscopy?
-
Consider the following two compounds. Monochlorination of one of these compounds produces twice as many stereoisomeric products as the other. Draw the products in each case, and identify which...
-
Consider the following two compounds. When treated with NaOH, one of these compounds forms an epoxide quite rapidly, while the other forms an epoxide very slowly. Identify which compound reacts more...
-
A solid of constant density is bounded below by the plane z = 0, on the sides by the elliptical cylinder x 2 + 4y 2 = 4, and above by the plane z = 2 - x. a. Find x and y. b. Evaluate the integral...
-
Write an executive summary on "Fracking revolution puts America on course to export oil"
-
Find bases such that this matrix represents the identity map with respect to those bases. 13 4' -1 1 4 2.
-
On what condition would you invest in a company that pays no dividend?
-
Two loudspeakers, A and B (Fig. 16.40), are driven by the same amplifier and emit sinusoidal waves in phase. Speaker B is 2.00 m to the right of speaker A. The frequency of the sound waves produced...
-
31.2 sin 0 2 cos 20 39. 2 cos 20 = 1 - cos 2
-
Suppose that in a production of spark plugs the fraction of defective plugs has been constant at over a long time and that this process is controlled every half hour by drawing and inspecting two...
-
Based on your analysis in the previous problem, determine whether the following allene is expected to be chiral: Me , Me, c=c=c Me
-
Using the data in the following table, predict the sign and magnitude of ÎH° for each of the following reactions. In each case, identify whether the reaction is expected to be endothermic...
-
The United States ranks ninth in the world in per capita chocolate consumption; the average American eats 9.5 pounds of chocolate annually (Forbes, July 22, 2015). Suppose you are curious whether...
-
Please be mindful that your paper will need an executive summary, equal to an average of 10% of your total paper word count. in your summary you will need to discuss your paper three sections (what...
-
3 || Consider a soap bubble of dia meter urface tension coefficient pressure is 3mm. If the s sur! is 0.072 N/m and the external O Pagage, what is Pressure ? what is the bubble's internal gage
-
4. Helium has a molecular weight of 4.003. what is the weight of 2 cubic meters of helium at atmosphere and 20c.
-
5 as a Express standard atmospheric pressure head, in meters of Ethylene glycol water Methanol. @ Air
-
Kayak Company budgeted the following cash receipts (excluding cash receipts from loans received) and cash payments (excluding cash payments for loan principal and interest payments) for the first...
-
Suppose you are given a weighted graph, G, with n vertices and m edges, such that the weight of each edge in G is a real number chosen independently at random from the interval [0, 1]. Show that the...
-
Presented below are income statements prepared on a LIFO and FIFO basis for Kenseth Company, which started operations on January 1, 2024. The company presently uses the LIFO method of pricing its...
-
Show how you would prepare each of the following carboxylic acids through a Grignard synthesis: (a) (b) (c) (d) 4-Methylbenzoic acid (e) Hexanoic acid OH OH
-
(a) Which of the carboxylic acids in Practice Problem 17.6 could be prepared by a nitrile synthesis as well? In problem 17.6 (b) Which synthesis, Grignard or nitrile, would you choose to prepare OH ...
-
When maleic acid is heated to 200 8C, it loses water and becomes maleic anhydride. Practice Problem 17.8 Fumaric acid, a diastereomer of maleic acid, requires a much higher temperature before it...
-
ZZZ Inc. plans to sell it's business and has used capitalization of earnings as a valuation method. with a stable cash flows of P2Million for the last 5 years. Forecast shows that similar level of...
-
The December 31, 20X1, physical inventory of Snowville Company appropriately included $6,300 of merchandise purchased on account that was not recorded in purchases until January 20X2. What effect...
-
Please notice that each line item has a respective section for Debit and Credit. If found appropriate, please ensure they are filled out. You do not have to give a thorough explanation; however,...

Study smarter with the SolutionInn App


