a. Evaluate the electronic partition function for atomic Si at 298 K given the following energy levels:
Question:
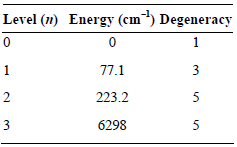
b. At what temperature will the n = 3 energy level contribute 0.100 to the electronic partition function?
Transcribed Image Text:
Level (1) Energy (cm) Degeneracy 77.1 223.2 5 6298 5 3. 2. 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a b Focusing on the contribution to q E from ...View the full answer

Answered By

Shubhradeep Maity
I am an experienced and talented freelance writer passionate about creating high-quality content. I have over five years of experience working in the field and have collaborated with several renowned companies and clients in the SaaS industry.
At Herman LLC, an online collective of writers, I generated 1,000+ views on my content and created journal content for 100+ clients on finance topics. My efforts led to a 60% increase in customer engagement for finance clients through revamping website pages and email interaction.
Previously, at Gerhold, a data management platform using blockchain, I wrote and published over 50 articles on topics such as Business Finance, Scalability, and Financial Security. I managed four writing projects concurrently and increased the average salary per page from $4 to $7 in three months.
In my previous role at Bernier, I created content for 40+ clients within the finance industry, increasing sales by up to 40%.
I am an accomplished writer with a track record of delivering high-quality content on time and within budget. I am dedicated to helping my clients achieve their goals and providing exceptional results.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Evaluate the electronic partition function for atomic Fe at 298 K given the following energy levels. Level (1) Energy (cm) Degeneracy 415.9 704.0 888.1 3 978.1 2. 3. 4.
-
NO is a well-known example of a molecular system in which excited electronic energy levels are readily accessible at room temperature. Both the ground and excited electronic states are doubly...
-
Why is the electronic partition function generally equal to the degeneracy of the ground electronic state?
-
All organizations incur non value added costs. Eliminating or reducing non value added costs is the responsibility of managers. Using the Activity Based Management approach and focusing on the...
-
In Exercises 1-2, find the domain of the function and discuss the behavior of f near any excluded x-values. 1. f (x) = 1 / x - 1 2. f (x) = 4 / x + 3
-
1. As completely as possible, sketch the value chain for Pandora from the production of content to the listener. 2. How do horizontal and vertical conflict impact Pandora? 3. How does Pandora add...
-
The Astro Co., a wholesaler of food products, budgeted the following sales for the months shown below: All merchandise is marked up to sell at its invoice cost plus 25 percent. Merchandise...
-
Rand Medical manufactures lithotripters. Lithotripsy uses shock waves instead of surgery to eliminate kidney stones. Physicians Leasing purchased a lithotripter for $3,000,000 and leased it to...
-
Given vectors u and u = (9.7). v= (2,0) (a) 4u=>
-
What important capability is lacking in a markup language?
-
Imagine performing the coin-flip experiment of Problem P29.17, but instead of using a fair coin, a weighted coin is employed for which the probability of landing heads is two-fold greater than...
-
Rhodopsin is a biological pigment that serves as the primary photoreceptor in vision (Science 266 [1994]: 422). The chromophore in rhodopsin in retinal, and the absorption spectrum of this species is...
-
The cable of a crane is lifting a 750 kg girder. The girder increases its speed from 0.25 m/s to 0.75 m/s in a distance of 3.5 m. a. How much work is done by gravity? b. How much work is done by...
-
Rearrange this equation to isolate c. a = b -(-) C =
-
Given the following data, what is the value of gross profit as determined by the average cost method? Sales revenue 300 units at $15 per unit Purchases 240 units at $10 per unit Beginning inventory...
-
Factor. 4c + 15c+9
-
The current price of a stock is $47, and three-month call options with a strike price of $50 currently sell for $4.70. An investor who feels that the price of the stock will increase is trying to...
-
What is motivation, discuss factors of motivation. Also, discuss Maslow's hierarchy of needs in detail (with examples).
-
A sample of 0.2140 g of an unknown monoprotic acid was dissolved in 25.0 mL of water and titrated with 0.0950 M NaOH. The acid required 27.4 mL of base to reach the equivalence point. (a) What is the...
-
A company produces earbuds. The revenue from the sale of x units of these earbuds is R = 8x. The cost to produce x units of earbuds is C = 3x + 1500. In what interval will the company at least break...
-
Predict the products of each of the following reactions: a. b. c. d. e. f. g. h. i. 1) BH3 THF 2) H2O2, NaOH Pt
-
The rate at which two methyl radicals couple to form ethane is significantly faster than the rate at which two tert-butyl radicals couple. Offer two explanations for this observation.
-
There are only two stereo-isomers of 1, 4-dimethylcyclohexane. Draw them, and explain why only two stereo-isomers are observed.
-
Acaba de contratar a un nuevo cliente que lleva ms de un ao en el negocio y que ha estado utilizando Excel para hacer un seguimiento de su actividad empresarial mensual. Qu 2 funciones de QuickBooks...
-
Barrhaven-brew, a local Ottawa micro-brewery, recently merged with a bottle making plant in New York to complement its Barrhaven brewing facilities. Barrhaven-brew has two divisions: Bottle Making...
-
De qu dos formas puedes crear una plantilla personalizada para un proyecto? Duplicar una plantilla de QuickStart Importar una plantilla Descargar proyectos desde la pestaa de proyectos compartidos...

Study smarter with the SolutionInn App


