Evaluate the electronic partition function for atomic Fe at 298 K given the following energy levels. Level
Question:
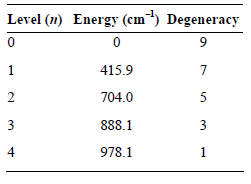
Transcribed Image Text:
Level (1) Energy (cm) Degeneracy 415.9 704.0 888.1 3 978.1 2. 3. 4.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
q ge 9e 7eB4159 cm 0 1 SeB7040cm 4159 cm ...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
a. Evaluate the electronic partition function for atomic Si at 298 K given the following energy levels: b. At what temperature will the n = 3 energy level contribute 0.100 to the electronic partition...
-
NO is a well-known example of a molecular system in which excited electronic energy levels are readily accessible at room temperature. Both the ground and excited electronic states are doubly...
-
Evaluate the translational partition function for H 2 confined to a volume of 100 cm 3 at 298 K. Perform the same calculation for N 2 under identical conditions. (Do you need to reevaluate the full...
-
On April 30, the end of the first month of operations, Joplin Company prepared the following income statement, based on the absorption costing concept: Joplin Company Absorption Costing Income...
-
In Exercises 51-54, a. determine the value that the function f approaches as the magnitude of x increases. Is f (x) greater than or less than this value when b. x is positive and large in magnitude...
-
Describe intermodal transportation and list the different combinations used to distribute products and the benefits of using this mode of transportation.
-
All sales of Dunns Building Supplies (DBS) are made on credit. Sales ar billed twice monthly, on the tenth of the month for the last half of the prior months sales and on the 20th of the month for...
-
Synergy and Dynaco are the only two firms in a specific high-tech industry. They face the following payoff matrix as they decide upon the size of their research budget: a. Does Synergy have a...
-
Tiberius Manufacturing is considering two alternative investment proposals with the following data: Investment Useful life Estimated annual net cash inflows for 5 years Proposal X $10,600,000 5 years...
-
Consider a narrow b and FM signal approximately defined by s (t) = A c cos (2?? c t) ? ?A c sin (2?? c t) sin (2?? m t) (a) Determine the envelope of this modulated signal. What is the ratio of the...
-
Imagine performing the coin-flip experiment of Problem P29.17, but instead of using a fair coin, a weighted coin is employed for which the probability of landing heads is two-fold greater than...
-
Rhodopsin is a biological pigment that serves as the primary photoreceptor in vision (Science 266 [1994]: 422). The chromophore in rhodopsin in retinal, and the absorption spectrum of this species is...
-
Indicated on the graph in Fig. 6.22. Write the coordinates of the indicated point. Fig. 6.22. 5- 26 4. 34 3- |25 33 32 28 27 -5-4 3 2 1, 2 3 4 5 31 -2 30 -3 29 -4 -5
-
Yard Professionals Incorporated experienced the following events in Year 1, its first year of operation: 1. Performed services for $27,500 cash. 2. Purchased $5,000 of supplies on account. 3. A...
-
SALZA TECHNOLOGY CORPORATION Annual Income Statements (in $ Thousands) 2019 Net sales $375 Less: Cost of goods sold 225 Gross profit 150 Less: Operating expenses 46 Less: Depreciation 25 Less:...
-
Write a Java program that checks if a string is a palindrome using a Deque. Your program will output "Yes" if it is a palindrome and "No" if it is not. You must solve this using a Deque class which...
-
State Motor Vehicles and Transportation Agency (SMVTA) conducts a study and concludes that most accidents involving pass3enger buses are caused by bus drivers who are under the influence of alcohol...
-
ces Coastal's balance sheet for a recent year revealed the following information: Current assets Noncurrent assets Noncurrent liabilities Stockholders' equity Required: $ 890,000 470,000 400,000...
-
A sample of 0.1687 g of an unknown monoprotic acid was dissolved in 25.0 mL of water and titrated with 0.1150 M NaOH. The acid required 15.5 mL of base to reach the equivalence point. (a) What is the...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
Consider the structure of the following compound: (a) When this compound is treated with bromine under conditions that favor monobromination, two stereoisomeric products are obtained. Draw them, and...
-
Name one element that you would expect to exhibit bonding properties similar to boron. Explain?
-
Nicotine is an addictive substance found in tobacco. Identify the hybridization state and geometry of each of the nitrogen atoms in nicotine: C-H N. C- H. Nicotine z: I I-O
-
Comparative Balance Sheets June 30, 2019 and 2018 2019 2018 Assets Cash Accounts receivable, net Inventory Prepaid expenses Total current assets Equipment Accum. depreciation-Equipment Total assets...
-
A company issued 10-year, 7.75% bonds with a face value of $100,000. The company received $97,817 for the bonds. Using the straight-line method of amortization, the amount of interest expense for the...
-
Single Plantwide Factory Overhead Rate Scrumptious Snacks Inc. manufactures three types of snack foods: tortilla chips, potato chips, and pretzels. The company has budgeted the following costs for...

Study smarter with the SolutionInn App


