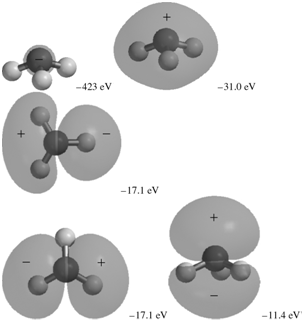
The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are
Question:
Transcribed Image Text:
-31.0 eV -423 eV -17.1 ev -17.1 eV -11.4 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The MO with an energy of 423 eV corresponds to the 1s AO on N It is a loca...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The occupied MOs of ethene are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
The occupied MOs of hydrogen cyanide are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as...
-
Indicate the type of solute-solvent interaction (Section 11.2) that should be most important in each of the following solutions: (a) CCl4 in benzene (C6H6), (b) Methanol (CH3OH) in water, (c) KBr in...
-
What is the is the effect of abolition of an import quota on (i) national saving, (ii) domestic investment, (iii) NCO, (iv) the real exchange rate, and (v) net exports?
-
(a) Use a graphing utility to complete the table. (b) Make a conjecture about the relationship between sin and sin(180 ). 0 20 0 40 60 80 sin e sin(180 0)
-
Data for Betsy Strand are presented in BE10-5. Prepare the employer's journal entries to record (a) Betsy's pay for the period and (b) The payment of Betsy's wages. Use January 15 for the end of the...
-
The internal audit department of Sachem Manufacturing Company is considering buying computer software that will aid in the auditing process. Sachem's financial and manufacturing control systems are...
-
As of December 31 of the current year, Petersen Corporation has prepared the following information regarding its liabilities and other obligations: Notes payable, of which $12,000 will be repaid...
-
Exercise 22-27A (Algo) Merchandising: Preparing a cash budget LO P4 Use the following information to prepare the July cash budget for Acco Company Ignore the "Loan activity" section of the budget a....
-
Beech Corporation is a merchandising company that is preparing a master budget for the third quarter of the calendar year. The companys balance sheet as of June 30th is shown below: Beechs managers...
-
Use the VSEPR method to predict the structures of the following: a. PF 3 b. CO 2 c. BrF 5 d. SO 2 3
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
Shareholders' funds as at 17 March 2012 were \( 5,629\) million (19 March 2011: \( 5,424\) million), an increase of \( 205\) million. This is mainly attributable to the continued profitable growth of...
-
Suppose you are driving at 60 mi/h (26.8 m/s) when you let up on the gas, step on the brakes and accelerate at a constant value of -1.8 m/s for 4.5 s. At the end of that time, what will be the...
-
ending inventory consists of 290 units from the January 30 purchase, 5 units from the January 20 purchase, and 20 uni from beginning inventory. Date January 1 January 10 Activities Beginning...
-
Suppose that you are the assistant manager of AW-tronics , a large privately held online retailer that currently uses 27 unskilled workers and 16 semiskilled workers at its warehouse to box and ship...
-
a. Competitive Environment - Identify Big Rock's main competitors (micro/macro) and determine what they are doing that might pose a threat or an opportunity for Big Rock. What are the competitor's...
-
Vulcan Flyovers offers scenic overflights of Mount Saint Helens. Data concerning the company's operations in July appear below: Vulcan Flyovers Operating Data For the Month Ended July 31 Flights (g)...
-
a. What products (including stereoisomers if applicable) would be formed from the reaction of 3-bromo-2-methylpentane with HO - under S N 2 /E2 conditions and under S N 1/El conditions? b. Answer the...
-
Reduction in sales All of the above 29. Belt of an electric motor is broken, it needs a. Corrective maintenance b. Scheduled maintenance c. Preventive maintenance d. Timely maintenance. 30. The...
-
The reaction mechanism involves an intermediate A. Deduce the rate law for the reaction. AA+A (fast) A+B P (slow)
-
A reaction 2 A P has a second-order rate law with k = 3.50 10 4 dm 3 mol 1 s 1 . Calculate the time required for the concentration of A to change from 0.260 mol dm 3 to 0.011 mol dm 3 .
-
Consider the dimerization 2 A A 2 , with forward rate constant k a and backward rate constant k b . (a) Derive the following expression for the relaxation time in terms of the total concentration of...
-
Which of the following is an internal control?Select one:a.A review of receipts against deposits.b.A monthly trial balance which ensures that all accounts are properly balanced.c.Keeping of petty...
-
Which of the following is not a motivating factor to move a firm's operations overseas? a. Lower production costs. b. High overseas taxes on non-repatriated earnings. c. Avoidance of tariffs. d....
-
Portal Doors sells steel entry doors for $285 each. The firm has fixed costs of $380,000 and variable costs per unit of $125. What is Portal's break-even point in sales dollars? Select answer from...

Study smarter with the SolutionInn App


