The occupied MOs of hydrogen cyanide are shown next along with the MO energies. Indicate which AOs
Question:
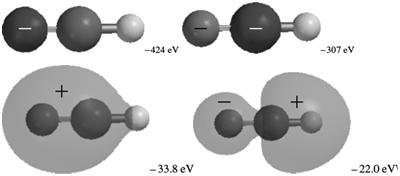
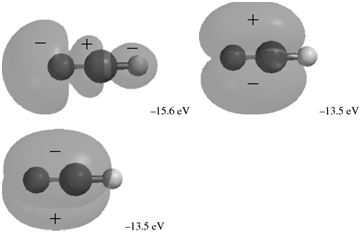
Transcribed Image Text:
-424 ev -307 eV -33.8 eV -22.0 eV メ -15.6 eV -13.5 eV -13.5 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The MOs corresponding to the energies 424 eV and 307 eV are 1s AOs on N and C respective...View the full answer

Answered By

AJIN kuriakose
I have completed B.Tech in Electrical Engineering & Masters in Power & Control From one of the best universities in India. I got the 99.05 percentile in the Gate Electrical Engineering Exam. I can Help students solving assignments in Electrical subjects like Power Electronics, Control system, Analog, Network Theory & Engineering Mathematics. Clear your fundamentals and develop problem-solving skills and analytical skills to crack the exam.
Get guidance and the opportunity to learn from experienced...
I can provide tuition for Electrical engineering subjects (Power Electronics, Digital electronics, Network Theory, Control System & Engineering Mathematics). The toughest subject of Electrical engineering can be made simple in online classes...
I can also solve it.
1 .I can help you with your assignments or exams or quiz or tutoring.
2. Very strict to the deadlines.
Message me for any help in assignments, live sessions. I am here to help students for all assignments, tests and exams and I will make sure you always get _95% In your subject.
Contact me in solution inn for any help in your semester, projects and for many more things . Also feel free to contact me through solution inn and for any advise related to tutoring and how it works here.thank you.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
The occupied MOs of ethene are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
Indicate the type of solute-solvent interaction (Section 11.2) that should be most important in each of the following solutions: (a) CCl4 in benzene (C6H6), (b) Methanol (CH3OH) in water, (c) KBr in...
-
2. There are different kinds of statistical analyses, and you will need to use one that is best suited to the data available and the information you need to collect in the given scenario (Good Days)....
-
Determine whether the statement is true or false. Justify your answer. 1. Determine whether the statement is true or false. Justify your answer. In each of the four quadrants, the signs of the secant...
-
Woode Corporation issued $400,000, 7%, 20-year bonds on January 1, 2017, for $360,727. This price resulted in an effective-interest rate of 8% on the bonds. Interest is payable annually on January 1....
-
As the manager of the external audit team, you realize that the embedded audit module only writes "material" invoices to the audit file for the accounts receivable confirmation process. You are...
-
The Village of Seaside Pines prepared the following enterprise fund Trial Balance as of December 31, 2017, the last day of its fiscal year. The enterprise fund was established this year through a...
-
On December 31, Lujack Company held the following short-term available-for-sale securities. Lujack had no short-term investments prior to the current period. Prepare the December 31 year-end...
-
Tempest Manufacturing has 8 million shares of common stock outstanding. The current share price is $73, and the book value per share is $7. Tempest Manufacturing also has two bond issues outstanding....
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
Use the geometrical construction shown in Example Problem 24.8 to derive the electron MO levels for cyclobutadiene. What is the total energy of the molecule? How many unpaired electrons will the...
-
Find the area of the polygon. 4 7
-
The sales of the cashmere jumpers are seasonal. Projected sales for July - Dec (the first six months of the year) are 18,000 units. Projected sales for Jan - Jun (the second six months) are 22,000...
-
Smith-Kline Company maintains inventory records at selling prices as well as at cost. For 2024, the records indicate the following data: Beginning inventory Purchases Freight-in on purchases Purchase...
-
Matt wants to ensure that critical network traffic from systems throughout his company is prioritized over web browsing and social media use at this company. What technology can he use to do this?
-
Kubin Company's relevant range of production is 20,000 to 23,000 units. When it produces and sells 21,500 units, its average costs per unit are as follows: Average Cost per Unit $ 8.00 Direct...
-
Cane Company manufactures two products called Alpha and Beta that sell for $165 and $130, respectively. Each product uses only one type of raw material that costs $8 per pound. The company has the...
-
The pK a of acetic acid in water is 4.76 (Section 1.17). What effect would a decrease in the polarity of the solvent have on the pK a ? Why?
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
At 518C, the rate of decomposition of a sample of gaseous acetaldehyde, initially at a pressure of 363 Torr, was 1.07 Torr s 1 when 5.0 per cent had reacted and 0.76 Torr s 1 when 20.0 per cent had...
-
The ClO radical decays rapidly by way of the reaction, 2 ClO Cl 2 + O 2 . The following data have been obtained: Determine the rate constant of the reaction and the half-life of a ClO radical....
-
The rate law for the reaction in Exercise 22.2a was reported as d[C]/dt = k[A][B][C]. Express the rate law in terms of the reaction rate; what are the units for k in each case? Data in Exercise 22.2a...
-
Which of the following statements is correct? Group of answer choices The ePayment Code applies to every ADI subscriber in Australia. There is no such thing as the ePayment Code All of these answers...
-
Accounting I16) The Chung Chung Chung Corporation uses a periodic inventory system. Thecompany has a beginning inventory of 1,250 units at $15 each on January 1. Chungchung chung purchases 1,500...
-
Iverson, Inc. has $1,296,000 of sales revenue, $706,000 of variable costs, and $659,000 of fixed costs in 2023. Assuming the contribution margin percentage stays the same in 2024, what will the break...

Study smarter with the SolutionInn App


