a. One microliter of 0.1 g/mL 0.1 g/mL benzene is injected into a gas chromatograph using splitless
Question:
a. One microliter of 0.1 μg/mL 0.1 μg/mL benzene is injected into a gas chromatograph using splitless injection. How much benzene is loaded onto the column?
b. How much benzene is loaded onto the column if split injection with a split ratio of 100:1 100:1 is used?
c. If both peak widths are 1 s, 1 s, < what is the mass of carbon entering the detector for splitless and split injection in (a) and (b)?
d. Based on the detection limits in Table 24-3, would a flame ionization detector be sensitive enough to detect the splitless and split injections?
Table 24-3
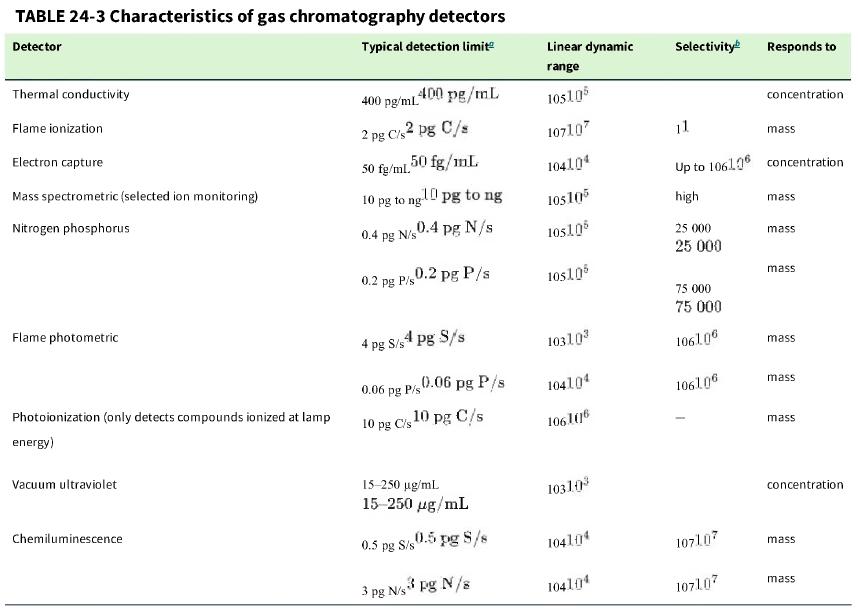
Transcribed Image Text:
TABLE 24-3 Characteristics of gas chromatography detectors Detector Typical detection limit Thermal conductivity Flame ionization Electron capture Mass spectrometric (selected ion monitoring) Nitrogen phosphorus Flame photometric Photoionization (only detects compounds ionized at lamp energy) Vacuum ultraviolet Chemiluminescence 400 pg/mL 2 pg C/s2 pg C/s 50 fg/mL. 10 pg to 0.06 400 pg/mL 50 fg/mL 0.4 pg N/s 0.2 pg P/s 0.2 pg P/s 0.4 pg N/s 4 pg 8/s4 pg S/s pg 3 gl[ pg to ng ng pg 10 pg C/s1 P/s 0.06 pg P/s 10 pg C/s 15-250 µg/mL 15-250 µg/mL 0.5 pg S/s0.5 pg S/s N/s3 pg N/s Linear dynamic range 105105 107107 104104 105105 105105 105105 10310³ 104104 1061016 10310³ 1041014 104104 Selectivity 25 000 25 000 11 Up to 106106 concentration high 75 000 75 000 1061.06 106106 107107 Responds to 107107 concentration mass mass mass mass mass mass mass concentration mass mass
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a The amount of benzene loaded onto the column can be calculated using the equation Amount of ben...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Peak areas and relative detector responses are to be used to determine the concentration of the five species in a sample. The area normalization method described in Problem 32-20 is to be used. The...
-
A chunk of ice of mass ml = 100 g at a temperature tx = 0 C was placed in a calorimeter in which water of mass m1 = 100 g was at a temperature t1. Assuming the heat capacity of the calorimeter to be...
-
A gas chromatograph is an instrument that measures the amounts of various compounds contained in a sample by separating the various constituents. Because different components are flushed through the...
-
1. About 99 percent of the world's population lives outside Canada, but many Canadian companies, especially small businesses, still do not engage in global trade. Why not? Do you think more small...
-
Let the random variable D be defined as in Exercise 7, and let the random variable Q2 be defined by Eq. (11.3.30). a. Show that b. Explain why the random variable Q2/2 will have the 2 distribution...
-
Kasperov Corporation has an unlevered cost of equity of 12% and is taxed at a 40% rate. The 4-year forecasts of free cash flow and interest expenses are shown below. Free cash flow and interest...
-
From the following selected balances of Filo plc as at 31 March 2005 draw up (i) a detailed trading and profit and loss account for internal use, and (i) a profit and loss account for publication....
-
Entries for Disposition of Assets On December 31, 2010, Chrysler Inc. has a machine with a book value of $940,000. The original cost and related accumulated depreciation at this date are as follows....
-
JOHN joined in organization for a fortnight and he had conversation with RAM . JOHN was hired as company counselor; felt that he was fed up with ram's team of non-Performa. There had been two major...
-
1. Did Ed commit an act discreditable to the profession? Explain. 2. Does the case have any bearing on Ed's ability to provide competent services to his client and serve the public interest? Why or...
-
Efficiency with HPLC Teaching Assistant, an Excel simulation of high performance liquid chromatography. Download the Excel file from SaplingPlus, macmillanlearning.com, or the link in reference...
-
Match descriptions 1-5 1-5 with the band broadening terms in the second list. 1. Depends on radius of open tubular column 2. Not present in an open tubular column 3. Depends on length and radius of...
-
Vintage Automobiles of Philadelphia, Inc., was formed on January 1, 20X8, when Vintage issued its common stock for $300,000. Early in January, Vintage made the following cash payments: a. $150,000...
-
A clinic uses doctors and nurse practitioners optimally and is servicing the maximum number of patients given a limited annual payroll. The last doctor hired treated 1200 extra patients in a year,...
-
The state of north Arizona had 11,450 food service business in 2017 a dentist rate of 0.84 per 1000 people and a total smoker percentage of 17.6. Recent changes in public health policy have led to...
-
The following program contains no syntax errors. As it executes, it will create one or more processes. Show the hierarchy of processes and what values each variable has in each of these processes at...
-
Assessment 1 - Short Answer Questions BSBMKG434 Promote products and services Company : Bounce Fitness Question 1 The table below includes parts of a typical action plan used for business promotional...
-
An archer fires an arrow straight up into the air with a speed vo = 50.1 m/s. Neglect air resistance. In the drawing, for purposes of visualization, the trajectory of the arrow has been give a small...
-
Grete Corp. had the following foreign currency transactions during 2013: Purchased merchandise from a foreign supplier on January 20, 2013, for the U.S. dollar equivalent of $60,000 and paid the...
-
C- Consider the following scenario:- A supermarket needs to develop the following software to encourage regular customers. For this, the customer needs to supply his/her residence address, telephone...
-
A Standard Reference Material is certified to contain 94.6 ppm of an organic contaminant in soil. Your analysis gives values of 98.6, 98.4, 97.2, 94.6, and 96.2 ppm. Do your results differ from the...
-
Nitrite (NO2) was measured by two methods in rainwater and unchlorinated drinking water. The results standard deviation (number of samples) are Sample source Gas chromatography Spectrophotometry Rain...
-
Should the value 216 be rejected from the set of results 192, 216, 202, 195, and 204?
-
Select all that apply On 2/1, Sophie Company sold merchandise on account to Lexie Company in the amount of $1,500. Lexie Company paid the amount due on 2/28. The journal entry that Sophie Company...
-
The Latin expression nemo dat quod non habet means: Group of answer choices the auditor must respect the client's desire for secure transmission of data. the audit client should not be expected to...
-
I need help with my accounting chapter 19 mastery, can someone upload it to the page?

Study smarter with the SolutionInn App


