Based on Figure 21-6, what are the processes involved in atomic absorption of a solution of a
Question:
Based on Figure 21-6, what are the processes involved in atomic absorption of a solution of a metal ion? In atomic emission?
Figure 21-6
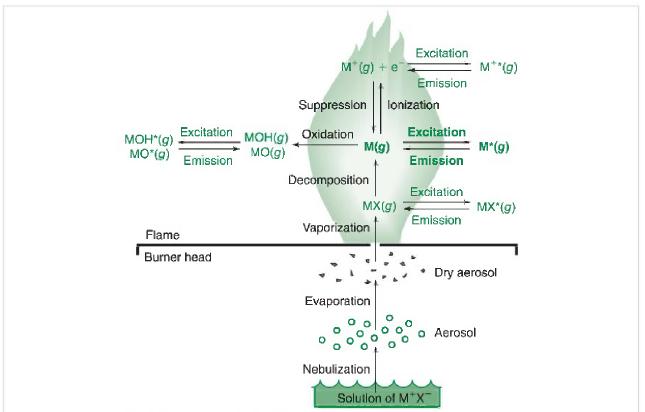
Transcribed Image Text:
Excitation MOH(g) MO(g) MOH*(g) MO*(g) Emission Flame Burner head M*(g) + e Suppression Oxidation M(g) Decomposition MX(g) Vaporization Evaporation lonization Nebulization Excitation Emission Excitation Emission Excitation Emission Solution of M*X M**(g) M*(g) MX*(g) Aerosol Dry aerosol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Solution The involved processes in atomic absorption of a solution of a metal ion are 1 In the absor...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
What are the processes involved in asset decommissioning/disposal? (abandonment planning, execution, and strategy) if the asset is a car? *emphasis of that in bold - I need about 3 pages worth...
-
What are the processes in cellular respiration?
-
Summarize the processes involved in project human resource management.
-
Where is the line in the sand the point where such behaviors are so destructive that you feel that the relationship needs to end?
-
Consider again the conditions of Exercise 12, and suppose that it is desired to estimate the value of = 5 40 + 1. Find an unbiased estimator Determine the value of and the M.S.E. of .
-
Cook asserts that an ERP and lean manufacturing through Continuous Improvement are not in conflict - that they are independent like the x- and y-axes. How do you react to that argument?
-
What is meant by the forensic-type fieldwork phase of an audit?
-
For Eckert Inc., variable manufacturing overhead costs are expected to be $20,000 in the first quarter of 2011, with $4,000 increments in each of the remaining three quarters. Fixed overhead costs...
-
Activity 4. Admission by purchase of Interest and Investment of Assets The capital accounts of Loida and Cris have balances of P150,000 and P110,000, respectively. Daria and Helen are to be admitted...
-
S&P Enterprises has provided data from the first three months of the year. The Controller has asked you to prepare the Cash Budget and the related Schedules for Expected cash collections and Payments...
-
What is the difference between luminescence, chemiluminescence, and bioluminescence?
-
Stern-Volmer plot. The table shows intensity and lifetime data for luminescence of the ruthenium complex in Figure 19-21 for a range of oxygen concentrations. a. Why does the luminescence intensity...
-
List insights that the statement of cash flows can provide to our analysis.
-
Explain the reasons why Interactive Media has become such a potent medium for doing effective advertising. 3. When you look at the whole picture of consumer market, and you look at all demographic...
-
Mr. Del Valle is 54 years old and plans to retire in 8 years. He wants to live at least 20 years after he retires. He has $125,000 in savings and expects to earn 7% annual interest. He expects to...
-
How can a company's EPS be influenced by changes in accounting policies? 13. How does the market react to changes in a company's EPS? 14. How can investors use EPS to estimate future earnings and...
-
In Fig. shows a 12.0 V battery and four uncharged capacitors C= 1.0 F, C2= 2.0 F, C3-3.0 F and C4= 4.0 F. If switch S is closed, what is the charge on each capacitor? What is the voltage across C? C...
-
How do interest rates affect the value of debentures? 12. What factors should investors consider when analyzing the risk and return of debentures? 13. How are debentures different from bonds and...
-
Opus, Incorporated, owns 90 percent of Bloom Company. On December 31, 2012, Opus acquires half of Bloom's $500,000 outstanding bonds. These bonds had been sold on the open market on January 1, 2010,...
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
Write the Ka and Kb reactions of NaHCO3.
-
Write the stepwise acid-base reactions for the following ions in water. Write the correct symbol (for example, Kb1) for the equilibrium constant for each reaction. a. b. H3NCH2CH2NH...
-
For the reaction 2A(g) + B(aq) + 3C(l) D(s) + 3E(g), the concentrations at equilibrium are found to be A: 2.8 103 pa B: 1.2 10-2 M C: 12.8 M D:16.5M E:3.6 104 Torr Find the numerical value of the...
-
What ingredients are needed to power a dynamo effect inside a planet? Group of answer choices A liquid iron core with a high density that is covered by a thick layer of liquid metallic hydrogen. A...
-
Physical mutagens include ______. Multiple choice question. neither X-rays nor UV light both X-rays and UV light UV light but not X-rays X-rays but not UV light
-
True or False: Percentage body fat can be measured by using calipers, bioelectrical impedance, dual-energy X-ray absorptiometry, underwater weighing, or air displacement. Ask Studybuddy Group of...

Study smarter with the SolutionInn App


