A gas is compressed at a constant pressure of 50 N/m from a volume of 10m...
Fantastic news! We've Found the answer you've been seeking!
Question:
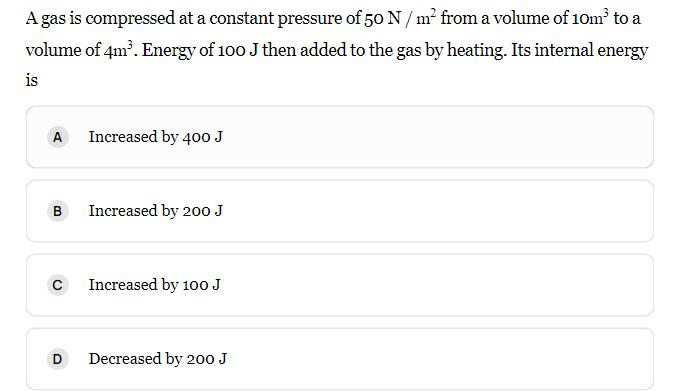
Transcribed Image Text:
A gas is compressed at a constant pressure of 50 N/m² from a volume of 10m³ to a volume of 4m³. Energy of 100 J then added to the gas by heating. Its internal energy is A B C D Increased by 400 J Increased by 200 J Increased by 100 J Decreased by 200 J A gas is compressed at a constant pressure of 50 N/m² from a volume of 10m³ to a volume of 4m³. Energy of 100 J then added to the gas by heating. Its internal energy is A B C D Increased by 400 J Increased by 200 J Increased by 100 J Decreased by 200 J
Expert Answer:
Answer rating: 100% (QA)
ANSWER C Increased by 100 J CALCULATION PV nRT P 50 N... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
A gas is compressed at a constant pressure of 0.800 atm from 9.00 L to 2.00 L. In the process, 400 J of energy leaves the gas by heat. (a) What is the work done on the gas? (b) What is the change in...
-
The function Eint = 3.50nRT describes the internal energy of a certain ideal gas. A sample comprising 2.00 mol of the gas always starts at pressure 100 kPa and temperature 300 K. For each one of the...
-
Plss answer the highlighted number SERVATION OF MASS AND ENERGY unknown flow rate; however, the water temperature increases from 13C to 24 C. Also, it is known that 1 kg of water will absorb 4.2 kJ...
-
Motion of Electrons in a Magnetic Field An electron of mass m and charge -e is moving through a uniform magnetic field B = (B, 0, 0) in vacuum. (Figure 1)At the origin, it has velocity (Uz, Vy, 0),...
-
The Frost Company has accumulated the following information relevant to its 2007 earnings per share. 1. Net income for 2007, $150,500. 2. Bonds payable: On January 1, 2007 the company had issued 10%,...
-
The radiator of a steam heating system has a volume of 15 L and is filled with superheated water vapor at 225 kPa and 230oC. Now the inlet and exit ports are closed. After a while the temperature of...
-
Taylor Company recently purchased a piece of equipment for \(\$ 2,000\) that will be paid for within 30 days after delivery. At what point will the event be recorded in Taylor's accounting system? a....
-
Nasenko Companys cost and production data for two recent months included the following: Required a. Separately calculate the rental cost per unit and the utilities cost per unit for both March and...
-
A1. Let C be the curve in the my-plane that connects (1,2,0) to (0, 0, 0) along y = 2:52 and then (0, 0, 0) to (1, 1, 0) along y = :53. (a) By recognizing that F (as, y, z} = (25cc:er 29, 23:23\"...
-
The plots below show sampling distributions of p-values for different values of power. Match the distribution to the power. Please note that for this problem, you must pick values of power for all...
-
3 . a . List and describe three types of deferred credits? b . Do they meet the definition of a liability? Of short or long term? c . Why do some accountants not consider them to be a liability? d ....
-
Of the four factors that may influence alliance performance: equity, learning and experience, nationality, and relational capabilities; which do you think is the most important, and which is the...
-
a 2 0 - year, 7 % semi - annual coupon bond ( R 1 0 0 0 par value ) is priced at a yield maturity of 1 2 % . The yield changes by 4 0 basis points. the effective duration of the bond is closest to
-
WACC Estimation The following table gives the balance sheet for Travellers Inn Inc. (TII), a company that was formed by merging a number of regional motel chains. Travellers Inn: (Millions of...
-
4.C. When integrating a rational expression, symbolically we write f N(x) dx. What is your first D(x) step if the degree of N(x) is greater than or equal to the degree of D(x)? x33x
-
4.26 A particle of mass m and initial velocity, collides elastically with a particle of unknown mass A coming from the opposite direction as shown at left below. After the collision m has velocity...
-
A factory manufactures two products, A and B. Each product requires the use of three machines, Machine I, Machine II, and Machine III. The time requirements and total hours available on each machine...
-
Dawson Companys balance sheet information at the end of 2019 and 2020 is as follows: Additional information: The company did not issue any common stock during 2020. Required : Next Level Fill in the...
-
Reconsider Prob. 10-31. Using EES (or other) software, solve this problem by the diagram window data entry feature of EES. Include the effects of the turbine and pump efficiencies and also show the...
-
An insulated piston-cylinder device initially contains 300 L of air at 120 kPa and 17C. Air is now heated for 15 min by a 200-W resistance heater placed inside the cylinder. The pressure of air is...
-
2-kg of saturated water vapor at 600 kPa are contained in a piston-cylinder device. The water expands adiabatically until the pressure is 100 kPa and is said to produce 700 kJ of work output. (a)...
-
Adjusting Entries and Financial Statements} You have the following unadjusted trial balance for Rangoon Corporation at December 31, 2018: At year-end, you have the following data for adjustments: a....
-
Comprehensive Problem: Reviewing the Accounting Cycle} Fodor Freight Service provides delivery of merchandise to retail grocery stores in northern Manitoba. At the beginning of 2018 , the following...
-
Cash-Basis and Accrual-Basis Income} Martin Sharp, who repairs lawn mowers, collects cash from his customers when the repair services are completed. He maintains an inventory of repair parts that are...

Study smarter with the SolutionInn App


