Crystallographers have a rough rule of thumb that non-H atoms occupy around 18 3 in many
Question:
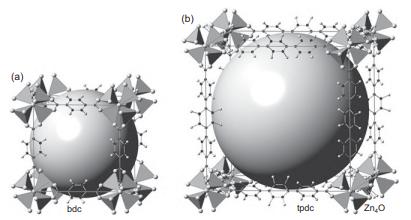
Crystallographers have a rough “rule of thumb” that non-H atoms occupy around 18 Å3 in many crystal structures. Based on this assumption, estimate the percent pore space in the tpdc MOF on the right of Figure 14.14.
Figure 14.14
Transcribed Image Text:
(a) bdc tpdc Zn0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The contents of the cell shown woul...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
After graduating from business school, George Clark went to work for a Big Six accounting firm in San Francisco. Because his hobby has always been wine making, when he had the opportunity a few years...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The Collectors Choice Inc. ( CCI), a luxury car import company, has an old warehouse at the Port of Miami, Florida, where it temporarily stores expensive sports cars and automotive parts that arrive...
-
For each polynomial function, complete the following in order. (a) Use Descartes rule of signs to determine the different possibilities for the numbers of positive, negative, and nonreal complex...
-
Find the wavelength of the K line in copper (Z = 29) if the wavelength of the K line in iron (Z = 26) is known to be equal to 193 pm.
-
An island economy produces only bananas and coconuts. The table gives the quantities produced and prices in 2018 and in 2019. The base year is 2018. Calculate nominal GDP in 2018 and nominal GDP in...
-
The following costs were incurred for the month of June for the Process A23 Department. Use this information to prepare a responsibility report for the month of June. Budget Actual Sales in units.....
-
Mira and Lemma are equal owners of an entity. Each contributed $25,000 cash to the entity. In addition, the entity obtains a loan of $100,000. The profits for the year are $30,000. Determine Mira and...
-
Determine the outstanding principal of the given mortgage. HINT [See Example 7.] (Assume monthly interest payments and compounding periods. Round your answer to the nearest cent.) a $100,000,...
-
Consider the PtS net listed in Table 1.6. The dehydrated form of MOF-11 has composition Cu 2 (ATC) where ATC is 1,3,5,7-adamantane tetracarboxylate and contains a Cu paddle-wheel unit analogous to...
-
The first peak in the powder diffraction pattern of a mesoporous MCM material with 30 pores recorded with a wavelength of 1.54 is at 2.2 2. Estimate the thickness of the silica walls. A hexagonal...
-
How does the duty of confidentiality differ from lawyerclient privilege?
-
Expand in two-dimensional Dv 1 Dt P Cartesian coordinates the following system of equations: (equation of motion) Vp+wv+f pcp Dt V.v=0 (mass conservation) kV0+ (energy equation) where =2uD: D is the...
-
A speeding car is pulling away from the police car. The police car is moving at 30 m/s. The speeding car is moving at 65 m/s. The radar gun in the police car emits an electromagnetic wave with a...
-
If a raindrop weighs 0.52mg on average and 5.13x10^5 raindrops fall on a lawn every minute, what mass (in kg) of rain falls on the lawn in 1.5h?
-
Stefani and raven are floating in space, and Stefani throws a dodgeball at raven. What will Stefani's motion look like after she has thrown the ball?
-
A 1490 kg Lamborghini Veneno can go from 0 mph to 60 mph (26.82 m/s) in 2.9 seconds. How much power does the engine provide?
-
Use the Kahl Medical Supplies data from P11-39B. The cash amounts for Interest Revenue, Salaries Expense, Interest Expense, and Income Tax Expense are the same as the accrual amounts for these items....
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
Give an example of two systems that are in equilibrium with respect to only one of two state variables.
-
Aerobic cells metabolize glucose in the respiratory system. This reaction proceeds according to the overall reaction 6O 2 (g) + C 6 H 12 O 6 (s) 6CO 2 (g) + 6H 2 O(l) Calculate the volume of oxygen...
-
Using acetylene as your only source of carbon atoms, outline a synthesis for 3-hexyne.
-
INSIDE JOB DOCUMENTARY GROUP ASSIGNMENT: (1) Summarize Part I (How We Got Here), Part II (The Bubble), Part III (The Crisis), Part IV (Accountability), Part V (Where We Are Now) (2) What is a...
-
Over the coming year, Ragwort's stock price will halve to $60 from its current level of $120 or it will rise to $240. The one-year interest rate is 12%. a. What is the delta of a one-year call option...
-
A political think tank wants to conduct a poll of likely voters in a state. If the think tank breaks the state into 20 groups with common characteristics and then randomly samples each group, what...

Study smarter with the SolutionInn App


