Name the following alcohols according to the IUPAC nomenclature system. Indicate stereochemistry (if any) and label the
Question:
Name the following alcohols according to the IUPAC nomenclature system. Indicate stereochemistry (if any) and label the hydroxy groups as primary, secondary, or tertiary.
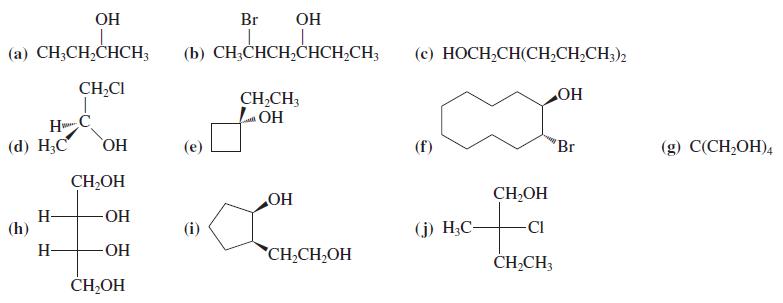
Transcribed Image Text:
OH Br OH (a) CH;CH,CHCH; (b) CH;CHCH,CHCH,CH; (c) HOCH,CH(CH,CH,CH;)2 CH,CI CH,CH, LOH HO H (d) Н,С HO CH,OH (f) 'Br (g) С(CH,ОH), CH,OH H- (h) HO OH (i) (j) H;C- CI H- -ОН CH,CH,OH ČH,CH; CH-OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
SNo IUPAC Name Type Of Alcohol Stereochemistry a Butan2ol secondary NA b 2bromohexan4ol s...View the full answer

Answered By

Bharat Singh Patel
I like teaching since my matriculation. That time i was teaching 6-8 class students with all subjects. I was very interested in teaching. Then after i have done my 10+2. I joined a coaching class. Where i was teaching mathematics and chemistry of 10th class student. After that i moved to Ewing Christian College, Allahabad for Graduation. then i taught chemistry only. then I clear IIT JAM to got admission in IIT Jodhpur for M.Sc. During M.Sc I have cleared CSIR-JRF With AIR 54 and GATE with AIR 114. In college time I always cleared doubts of classmates and juniors.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Classify the following alcohols as primary, secondary, or tertiary: (a) (b) (c) (d) OH
-
State whether each of the following alcohols is primary, secondary, or tertiary. a. b. c. d. -, ,
-
Classify the following amines as primary, secondary, or tertiary: (a) (b) (c) (d) (e) (f) H IN NH2 HN
-
Suppose that General Motors Acceptance Corporation issued a bond with 10 years until maturity, a face value of $1000, and a coupon rate of 7% (annual payments). The yield to maturity on this bond...
-
Describe the importance of managing stakeholder expectations.
-
Details of inheritance, 20 points. been defined: Assuming that the following classes have public class Couch extends Table { public void method1() { System.out.println(\"Couch 1\"); } } public class...
-
Which of the following techniques involves the creation and use of a pair of public and private keys? a. hashing c. symmetric encryption b. asymmetric encryption d. key escrow
-
FM Company provides fraud monitoring services. It employs four fraud specialists. Each specialist works an average of 200 hours a month. The company's controller has compiled the following...
-
compute for the following: Equivalent units for Materials is _____. Cost per unit for labour is _____. Closing WIP in USD is _____. Cost of sales under absorption costing is _____. Contribution...
-
A tall standpipe with an open top (see figure) has diameter d = 2.2 m and wall thickness t = 20 mm. (a) What height h of water will produce a circumferential stress of 12 MPa in the wall of the...
-
Which of the following isomeric carbocations is the most stable? CH,+ CH3 () (b) -CH3 CH3 () (d)
-
Write all products of the reaction of each of the alcohols in Problem 31 with concentrated H 2 SO 4 under elimination conditions. In Problem 31 OH () CH-CH-CH,O (b) CH;CHCH3 (c) CH;CH,CH,CH,OH OH (d)...
-
What message does an attestation report convey to readers?
-
What problem does developing logical ERs before physical ERs solve?
-
What is a physical interface?
-
Demonstrate the links among cost estimation, the WBS, and the project schedule.
-
What is a logical interface?
-
Your project is scheduled to last three months and cost $150,000. At the end of the first month, the project is 20% complete. What is the earned value (EV)?
-
Plastically deforming a metal specimen near room temperature generally leads to which of the following property changes? (A) An increased tensile strength and a decreased ductility (B) A decreased...
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
Calculate the difference in molar entropy (a) between liquid water and ice at -5C, (b) between liquid water and its vapour at 95C and 1.00 atm. The differences in heat capacities on melting and on...
-
A block of copper of mass 2.00 kg (Cp, m = 24.44 T K-I mol-1) and temperature OC is introduced into an insulated container in which there is 1.00 mol H20 (g) at 100C and 1.00 atm. (a) Assuming all...
-
At 20C, the density of a 20 per cent by mass ethanol-water solution is 968.7 kg m-3. Given that the partial molar volume of ethanol in the solution is 52.2 cm3 mol-1, calculate the partial molar...
-
Fake experts, please stay away Don't use Chat GPT or any A.I. tools to answer the question please do not copy from anywhere, and give a very brief explanation Answer only in text format, don't use...
-
Consider a firm listed on Toronto Stock Exchange (TSX) since 1990. Briefly explain why the stock price of the firm will increase when it announces that it will list as well on New York Stock Exchange...
-
1.Which Business Strategies theories both internal and external do you think are applicable in the business climate of 2017? 2.How does the Chaos theory change the outlook for business internally and...

Study smarter with the SolutionInn App


