Which of the following isomeric carbocations is the most stable? CH,+ CH3 () (b) -CH3 CH3 ()
Question:
Which of the following isomeric carbocations is the most stable?
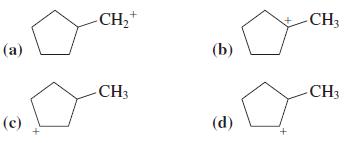
Transcribed Image Text:
CH,+ CH3 (а) (b) -CH3 CH3 (с) (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (13 reviews)
Tertiary Carbocations are the most stable Option a is a primary carbocat...View the full answer

Answered By

Saptarshi Paul
Education--
#Completed my Major in Chemistry in 2018
#Currently pursuing my Master's in Chemistry from IIT Kanpur
Tutoring Experience--
#Taught undergraduate level Chemistry to two students for a period of 6 months.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Which of the following reactions occurs most rapidly? Why? a. b. c. Br - H20 C(CH3)3 C(CH3)a (CH)a C(CH3)3 Br - H20 CH33 C(CH3)3
-
Which of the following compounds can be prepared by radical halogenation with little complication by formation of isomeric by-products? CI CI
-
Which of the following conformers of isobutyl chloride is the most stable Cl CH3 CH, H3C CH3 CH3 CH3 CI
-
On August 31, 2014, Nina Herrera borrowed $5,000 from Second State Bank. Herrera signed a note payable, promising to pay the bank principal plus interest on August 31, 2015. The interest rate on the...
-
Describe the main objective of the Information Distribution process.
-
Fix my code and make it complete: Question Write a class \"Account\" to represent a bank account with the following members: Account = {type, accountHolderName, creationDate, balance, category} Note...
-
Which of the following is a preventive control? a. penetration testing c. training b. patch management d. log analysis
-
The comparative historical-cost balance sheets of Majikstan Enterprises for 2010 and 2011 are reproduced below. The accounts are expressed in 000s of renges (MJRs). Required: What was the change in...
-
CABLE SOURCE ACQUIRED AN ASSET WITH A VALUE OF $3000000 AND A 4 YEAR LIFE. YEARLY CASH FLOWS ARE AS FOLLOWS YEARL 1 1,025,000 2 1,050,000 3 1,075,000 4 1,200,000 THE COST OF THE ASSET IS EXPECTED TO...
-
A software company is interested in improving customer satisfaction rate from the 75 % currently claimed. The company sponsored a survey of 281 customers and found that 221 customers were satisfied.
-
In this transformation, what is the best structure for A? (a) BrCH 2 CH 2 CH(CH 3 ) 2 (b) (c) (d) -, etone A CH;CH,C(CH3)2 H
-
Name the following alcohols according to the IUPAC nomenclature system. Indicate stereochemistry (if any) and label the hydroxy groups as primary, secondary, or tertiary. OH Br OH (a) CH;CH,CHCH; (b)...
-
What computations must be made if materials added in a department increase the number of units being processed in that department?
-
Evaluate project feasibility in terms of resources, funding, and facilities.
-
What System Elements comprise a systems Higher-Order Systems domain?
-
Describe and distinguish between qualitative and quantitative forecasting techniques. When should each of these forecasting techniques be used?
-
What are the two classes of OE domains?
-
Discuss the bill-of- quantities, function point, and three-point estimating methods, together with the merits and demerits of each.
-
A dislocation formed by adding an extra half-plane of atoms to a crystal is referred to as a (an) (A) Screw dislocation (B) Vacancy dislocation (C) Interstitial dislocation (D) Edge dislocation
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
The partial molar volumes of two liquids A and B in a mixture in which the mole fraction of A is 0.3713 are 188.2 cm3 mol-1 and 176.14 cm3 rnol-1 respectively. The molar masses of A and Bare 241.1 g...
-
Estimate the change in the Gibbs energy of 1.0 dm3 of water when the pressure acting on it is increased from 100 kPa to 300 kPa.
-
Calculate the change in the molar Gibbs energy of oxygen when its pressure is increased isothermally from 50.0 kPa to 100.0 kPa at 500 K.
-
thanks in advance... solve it but try to use a powerpoint with: .Introduction... .Background... (problem formulation and initial solution).... .Analysis... (decision variables, objective function,...
-
A researcher has a theory that people who read a set of facts on a green piece of paper are more likely to remember them than a subject who reads the facts from a pink piece of paper. In this...
-
A well-meaning public policy school Happy And Rigorous yet Riveting Institute for Studies (HARRIS) would like to know the effect of their MPP program on their students' future outcomes. They identify...

Study smarter with the SolutionInn App


