An ideal gas is characterised by the relation pV = NR T as in 1.2 where
Question:
An ideal gas is characterised by the relation pV = NR T as in § 1.2 where the pressure p (T, V) is a function of T and V, the temperature T (p, V) is a function of p and V and the volume (T, p) is a function of T and p. Calculate,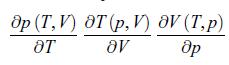
Transcribed Image Text:
др (T, V) OT (P, V) 8V (T,P) OT aV др
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
To find the partial derivatives we will use the relations pV NR T equation 1 p pT V eq...View the full answer

Answered By

Jayka Depante
As a tutor I am very friendly . Since last year I am giving IIT JAM tuition , among 9 students , 7 cracked this exam this year. I will try to teach every topic with practical examples. students are just like friends to me.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Principles Of Thermodynamics
ISBN: 9781108426091
1st Edition
Authors: Jean-Philippe Ansermet, Sylvain D. Brechet
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas is characterised by the relation pV = NRT where p is the pressure of the gas, V is the volume, T is the temperature, N is the number of moles of gas and R is a constant.a) Calculate the...
-
A gas is characterised by the enthalpy H(S, p) = C p T, where C p is a constant (called heat capacity and defined in 5.2), and by pV = NR T, where p is its pressure, V its volume, T its temperature...
-
Using java language and data structure In chapter 1 you learned how to merge two sorted lists and get a third list which is also sorted. Write a method named mergeUnsortedLists in an application,...
-
Record the following capital-related transactions for Private University plant funds: 1. Transfers of $250,000 are received from the current unrestricted fund for the purpose of funding the payment...
-
Civil Code 1719, subdivision (a) provides in part that any person who draws a check that is dishonored due to insufficient funds shall be liable to the payee for the amount owing upon the check and...
-
Golden Foods (GF) Ltd makes pies, pastries and pizzas, which it sells to retailers under its own brand-name and also supplies to a major supermarket chain. It has two sites: the bakery and its head...
-
1. What type of property was at the center of the dispute in this case? How did that property become involved in the dispute? 2. On what ground did the plaintiffs argue that the bank should not have...
-
A statistics teacher states the probability of a surprise quiz on any given day is 0.30. If quizzes are given independently each day, what is the probability there will be a surprise quiz on the next...
-
Rachel Cook is very concerned. Until recently, she has always had the golden touch, having successfully launched two start- up companies that made her a very wealthy woman. However, the timing could...
-
A basin contains Ns (t) moles of salt dissolved in Nw (t) moles of water. The basin receives fresh water at a constant rate in w. This water is assumed to be thoroughly mixed in the basin so that the...
-
Two of the Massieu functions are functions of the following state variables: 1. 2. The Massieu functions are obtained by performing Legendre transformations of the state function entropy S (U, V)...
-
Use VSEPR theory to predict the probable geometric structures of the molecules (a) O 2 XeF 2 ; (b) O 3 XeF 2 ; (c) OXeF 4 .
-
Why CEO of big companies put importance on human happiness? How does it relate to make a successful Human Resource Manager?
-
13. Which of the following is not part of the second line of defense? a. pH of the skin b. Cytokines c. Phagocytosis d. Fever 14. Cells infected with a virus produce glycoproteins that interfere with...
-
13. A chronic localized subcutaneous infection characterized by verrucoid lesions on the skin is a. candidiasis. b. leprosy. c. shingles. d. chromoblastomycosis. 14. The most important fungi that...
-
Based on Bite Toothpaste Bits: Creating a New Product Category Answer, 1. Analyze the possibility of success of the pill-based toothpaste category. 2. toothpaste packaged in sustainable plastic and...
-
Technology is changing the way accountants do their jobs. Find an article about an emerging accounting technology (for example, blockchain, data analytics tools, cryptocurrency, etc.). Post a link...
-
In the circuit shown in Fig. 7.81 v(t) = 56e200tV, t > 0 i(t) = 8e200tmA, t > 0 (a) Find the values of R and C. (b) Calculate the time constant Ï. (c) Determine the time required for the voltage...
-
Reduction in sales All of the above 29. Belt of an electric motor is broken, it needs a. Corrective maintenance b. Scheduled maintenance c. Preventive maintenance d. Timely maintenance. 30. The...
-
A 48.0 wt% solution of HBr in water has a density of 1.50 g/mL. (a) Find the formal concentration of HBr. (b) What mass of solution contains 36.0 g of HBr? (c) What volume of solution contains 233...
-
A solution contains 12.6 ppm of dissolved Ca(NO 3 ) 2 (which dissociates into Ca 2+ 2NO - 3 ). Find the concentration of NO - 3 in parts per million.
-
Ascorbic acid (vitamin C, page 354) reacts with I - 3 according to the equation Starch is used as an indicator in the reaction. The end point is marked by the appearance of a deep blue starch-iodine...
-
Mia Breen Corp. produces and sells wind-energy-driven engines. To finance its operations, Mia Breen issued $358,000 of 25-year, 10% callable bonds on May 1, 20Y5, at their face amount, with interest...
-
Sports Hats, Etc. has two product lines-baseball helmets and football helmets. Income statement data for the most recent year follow: Total Baseball Helmets Football Helmets Sales revenue $460,000...
-
HDFS 3350 DJ Richards wondered if he had adequate life insurance for his family if he were to pass away. He decided to complete the Life Insurance Worksheet. His current yearly income is $38,400. He...

Study smarter with the SolutionInn App


