An ideal gas is characterised by the relation pV = NRT where p is the pressure of
Question:
An ideal gas is characterised by the relation pV = NRT where p is the pressure of the gas, V is the volume, T is the temperature, N is the number of moles of gas and R is a constant.a) Calculate the differential dp (T, V)
b) Calculate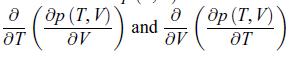
Transcribed Image Text:
8 ( 8p ( T, V ) ) OT 8Ꮴ and 8 ( Ꭷp ( T, V ) ᎥᏙ ᎧᎢ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
a To calculate the differential dp T V we first need to find the partial derivatives of p with respe...View the full answer

Answered By

Anik Kumar Bosu
I am expert in Maths, Physics, Chemistry and Biology. I also teach student from 3 years. Students becomes satisfied by my teaching. So, do not worry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Principles Of Thermodynamics
ISBN: 9781108426091
1st Edition
Authors: Jean-Philippe Ansermet, Sylvain D. Brechet
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas is characterised by the relation pV = NR T as in 1.2 where the pressure p (T, V) is a function of T and V, the temperature T (p, V) is a function of p and V and the volume (T, p) is a...
-
A gas is characterised by the enthalpy H(S, p) = C p T, where C p is a constant (called heat capacity and defined in 5.2), and by pV = NR T, where p is its pressure, V its volume, T its temperature...
-
Thermal Expansion of an Ideal Gas (a) The pressure p, volume V, number of moles n, and Kelvin temperature T of an ideal gas are related by the equation p V = nRT, where R is a constant. Prove that...
-
What are the "two main lines" of distortion of the view of Marx and what does Lenin think the proper view?
-
Record the following annuity and life income activities of Private University: 1. On July 1, 2010, R. W. Fields, emeritus professor of accounting, moved out of the state. Fields donated to the...
-
Civil Code 1719, subdivision (a) provides in part that any person who draws a check that is dishonored due to insufficient funds shall be liable to the payee for the amount owing upon the check and...
-
Outline the principal accounting and disclosure differences between IAS 16 and FRS 102, Section 17.1.
-
There is a database that contains a website's traffic data over a period of 30 days. The first table contains users' information including the user type (user, crawler, admin). The second table...
-
The following data show the number of rental cars in service for three rental car companies: Hertz, Avis, and Dollar over a three-year period.t Cars in Service (1000s) Company Year 1 Year 2 Year 3...
-
Zia Co. makes flowerpots from recycled plastic in two departments, Molding and Packaging. Zia uses the weighted average method, and units completed in the Molding department are transferred to the...
-
A basin contains Ns (t) moles of salt dissolved in Nw (t) moles of water. The basin receives fresh water at a constant rate in w. This water is assumed to be thoroughly mixed in the basin so that the...
-
Two of the Massieu functions are functions of the following state variables: 1. 2. The Massieu functions are obtained by performing Legendre transformations of the state function entropy S (U, V)...
-
What is the purpose of filing a lis pendens?
-
Using the internet and My Culinary Lab, define Energy Star, list some of the ways that a restaurant can use the Environmental Protection Agency's ENERGY STARSuperscript Guide for Cafes, Restaurants,...
-
4. Which of the following is not a common characteristic used in the selection of an antimicrobial drug? a. Selective toxicity b. Ease of delivery to the site of infection c. Ability to administer...
-
A project will increase revenue from $3.5 million to $4.5... A project will increase revenue from $3.5 million to $4.5 million. Wages are 60% of revenue. Maintenance on the machine will be $26,000...
-
Suppose a box of mass m slides toward a spring with a speed of vii, presses 1.20 mm into the spring, and then bounces away with a final speed of 17.0 m/s. Find the initial speed of the box. Take into...
-
Focuses on Organizational Behavior and the application of theory to the workplace. Take two concepts or theories covered in this course and apply the concepts or theories to your current or past...
-
The current through a 0.5-F capacitor is 6(1-e-t)A. Determine the voltage and power at t=2 s. Assume v(0) = 0.
-
Ex. (17): the vector field F = x i-zj + yz k is defined over the volume of the cuboid given by 0x a,0 y b, 0zc, enclosing the surface S. Evaluate the surface integral ff, F. ds?
-
A solution of NaOH was standardized by titration of a known quantity of the primary standard, potassium hydrogen phthalate The NaOH was then used to find the concentration of an unknown solution of H...
-
Write the names and abbreviations for each of the prefixes from 10 -24 to 10 24 . Which abbreviations are capitalized?
-
What is the formal concentration (expressed as mol/L = M) of NaCl when 32.0 g are dissolved in water and diluted to 0.500 L?
-
Reimel Furniture Company, Inc. incurred the following costs during 20x2. Direct material used Direct labor Manufacturing overhead $174,000 319,000 160,000 During 20x2, products costing $121,000 were...
-
Helix Corporation uses the weighted-average method in its process costing system. It produces prefabricated flooring in a series of steps carried out in production departments. All of the material...
-
Groucho, Harpo, and Chico form a partnership on January 1, 2021. Groucho contributes $270,000, Harpo $210,000, and Chico $120,000 to a business called Marx Brothers' Partnership. On a monthly basis,...

Study smarter with the SolutionInn App


