Suggest syntheses of these amino acids from the indicated starting materials: NH, Br a) PHCH,CHCO,H from PHCH,CHCO,H
Question:
Suggest syntheses of these amino acids from the indicated starting materials:
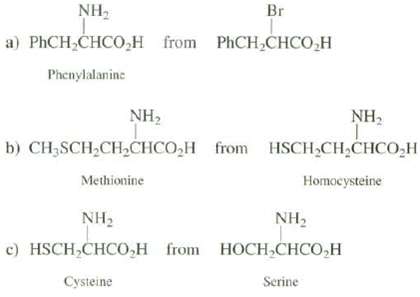
Transcribed Image Text:
NH, Br a) PHCH,CHCO,H from PHCH,CHCO,H Phenylalanine NH2 NH, b) CH3SCH2CHCHCO,H from HSCH CH,CHCO,H Methionine Homocysteine NH2 с) HSCH,CHCO,Н гom NH2 HОСH,CНCO,H Cysteine Serine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Br a PhCHCHCOH excess NH3 NH ...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral. , CH,CH,CH3 a)...
-
Propose syntheses of the following molecules from the indicated starting materials. Make use of any other reagents or solvents that you need. In some cases, there may be no alternative but to employ...
-
Show how Tagamet could be synthesized from the indicated starting materials. CH2OH CH CH SCH2CH2NH NHCH N CH CHS SCH CH3 +CH:NH2
-
Suggest two strategies for the company to minimise the impact of the strike on business operation?
-
In your own words, explain the difference between cost of goods manufactured and cost of goods sold.
-
Is it appropriate for an audit firm to function as a de facto adjunct to a clients accounting department? Why or why not? Which party or parties were primarily responsible for Deloitte being...
-
On July 1, 2020, West Company purchased for cash, eight \(\$ 10,000\) bonds of North Corporation to yield \(10 \%\). The bonds pay \(9 \%\) interest, payable on a semiannual basis each July 1 and...
-
Springsteen Co. had the following activity in its most recent year of operations. (a) Pension expense exceeds amount funded. (b) Redemption of bonds payable. (c) Sale of building at book value. (d)...
-
Neel, a single, self-employed taxpayer, is taking a trip to Singapore for 8 days. For 3 days he is attending a conference for work, and the rest is sightseeing. He has the following costs during his...
-
Financial information for American Eagle is presented in Appendix A at the end of the book. Required: 1. Calculate American Eagles percentage change in total assets and percentage change in net sales...
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
(a) Explain which of the following two products is formed when the reactant alkyl chloride reacts with sodium acetate in DMSO. (b) Explain which of the following two product alcohols is formed when...
-
It would be no surprise to most management students that as of May 2015, McDonalds was the sixth most valuable brand according to Forbes magazine. In terms of global restaurant brands it was the...
-
A train with a maximum speed of 105 km/h has an acceleration rate of 0.22 m/s 2 and a deceleration rate of 0.24 m/s 2 . If the train covers 1500 m while accelerating to maximum speed from rest, how...
-
Question 2 You are studying waiting times for hip replacement surgery in the NHS. You obtain a random sample of all patients who were waiting on January 1st, 2020 and follow it for 156 weeks. Your...
-
Fickel Company has two manufacturing departments-Assembly and Testing & Packaging. The predetermined overhead rates in Assembly and Testing & Packaging are $22.00 per direct labor-hour and $18.00 per...
-
How do sociological theories account for the role of technology in shaping modern societal norms and individual identities ?
-
Calculate inventory and cost of goods sold using FIFO, average-cost, and LIFO in a periodic inventory sy E6.6 (LO 2), AP Rusthe Inc. uses a periodic inventory system. Its records show the following...
-
For the spring-mass system shown in the given Fig.27, the frequency of oscillations of the block along the axis of the spring is (a) \(\frac{1}{2 \pi} \sqrt{\frac{k_{1}-k_{2}}{m}}\) (b) \(\frac{1}{2...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
Solve each equation. 8 2 =x + 3 5 + 23 11 -x+ 5 45 6x + 7 3
-
Pentalene is a most elusive molecule and has never been isolated. The pentalene dianion, however, is well known and quite stable. Explain. 12- Pentalene Pentalene dianion
-
Indole is an aromatic heterocycle that has a benzene ring fused to a pyrrole ring. Draw an orbital picture of indole. (a) How many ? electrons does indole have? (b) What is the electronic...
-
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1, 2, 4-triazole ring. Why is the ringaromatic? 1,2,4-Triazole ring N- NH2 N-N Ribavirin OH
-
Chapter 6 6. Complete the table by marking an X in the box that indicates that the control applies to the statement at the left in Table 2 or select "blank" if the control does not apply to the...
-
a. Is luu u always true or not always true? Explain the answer. OA. This is not true when components of the vector are negative. O B. 2 This is always true because lulu +42 + OC. This is not true for...
-
For the following graph of the shaded region that is bounded by y = x, x+y=6 and the x-axis: 5 3 2 Set up the integrals to calculate the area of the shaded region using dx and using dy. DO NOT...

Study smarter with the SolutionInn App


