A cumulene is a compound with three adjacent double bonds. Draw an orbital picture of a cumulene.
Question:
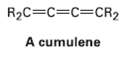
A cumulene is a compound with three adjacent double bonds. Draw an orbital picture of a cumulene. What kind of hybridization do the two central carbon atoms have? What is the geometric relationship of the sub-stituents on one end to the sub-stituents on the other end? What kind of isomerism is possible? Make a model to help see theanswer.
Transcribed Image Text:
R2C=C=C=CR2 A cumulene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
sp2 R R spsp o bond a bonds sp sp o bond bonds sp spsp o bond sp R3 R4 This simple...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw an orbital picture of furan to show how the molecule isaromatic. Furan :O:
-
Draw an orbital picture of thiazole. Assume that both the nitrogen and sulfur atoms are sp2-hyhridized, and show the orbitals that the lone pairs occupy.
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
The balance sheet for the Heir Jordan Corporation follows. Based on this information and the income statement in the previous problem, supply the missing information using the percentage of sales...
-
You are the Director of the Computer Center for Gaillard College and responsible for scheduling the staffing of the center, which is open from 8 a.m. until midnight. You have monitored the usage of...
-
Find the value of permutation. 4P4
-
1.5 Moscow to Tokyo. After spending a week in Moscow, you get an email from your friend in Japan. He can get you a very good deal on a plane ticket and wants you to meet him in Tokyo next week to...
-
The Berridge Company is a discount tire dealer that operates 25 retail stores in a metropolitan area. The company maintains a centralized purchasing and warehousing facility and employs a perpetual...
-
I am having trouble finding a solution to this problem. How would I prepare a vertical analysis of the balance sheet data for Nike for 2020. (Round percentages to 1 decimal place, e.g. 12.3%.. Assets...
-
Stockholders equity totaled $246,000 at the beginning of the year. During the year, net income was $36,000, dividends of $9,000 were declared and paid, and $30,000 of common stock was issued at par...
-
Terminal alkynes react with Br2 and water to yield bromo ketones. For example: Propose a mechanism for the reaction. To what reaction of alkenes is the processanalogous? -CECH Br2, H20 CH2Br
-
Reaction of acetone with D 3 O + yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of mercuric ion?catalyzed alkyne hydration, arid...
-
Air undergoes a normal shock. Upstream, \(T_{1}=35^{\circ} \mathrm{C}\), \(p_{1}=229 \mathrm{kPa}\) absolute, and \(V_{1}=704 \mathrm{~m} / \mathrm{s}\). Determine the temperature and stagnation...
-
What are the coupon rates for the following bonds: ( hint: find the annual coupon payment first, then divide it by the par value to find the coupon rate ) i ) Bond C: Par value = $ 1 0 0 0 ; years to...
-
1.If your VC firm tripled the fund in returns for a $125M fund, how much carry would the GPs get if you had a typical 2/20 fee structure?
-
Your broker called to recommend buying a specific stock because its P / E ratio is lower than the P / E ratio of the sector. Do you agree with this assertion? Provide reasons supporting your...
-
B. Astrid Inc. had the following balances in its ledger of accounts at the beginning of the year 2021: Beginning of the year balances Accounts receivable $380,000 Allowance for uncollectible accounts...
-
Explain how the concept of fugacity differs from ideal gas behavior in modeling phase equilibria, and illustrate its importance in predicting vapor-liquid equilibrium (VLE) for complex mixtures .
-
(a) Sketch \(v_{o}\) versus time for the circuit in Figure P2.18. The input is a sine wave given by \(v_{i}=10 \sin \omega t \mathrm{~V}\). Assume \(V_{\gamma}=0\). (b) Determine the rms value of the...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
Determine whether each redox reaction is spontaneous. (a) Fe(s) + Mg2+ (aq) (b) Fe(s) + Pb+ (aq) Fe2+ (aq) + Mg(s) 2+ Fe+ (aq) + Pb(s)
-
(a) Draw the structure of cis-CH3-CH=CH-CH2CH3 showing the pi bond with its proper geometry. (b) Circle the six coplanar atoms in this compound. (c) Draw the trans isomer, and circle the coplanar...
-
In pent-2-yne (CH3CCCH2CH3) there are four atoms in a straight line. Use dashed lines and wedges to draw a threedimensional representation of this molecule, and circle the four atoms that are in a...
-
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of the ones that do. (a) CH3CH=CHCH3 (b) CH3-C¡C-CH3 (c) CH2=C(CH3)2 (d) (e) (f) CH3-CH=N-CH3...
-
CK Ltd manufactures 1GB flash drives. The maximum production capacity is 200,000 units per month. The information related to the selling price and other cost data are as follows: Sales price per unit...
-
E9-7 (Algo) Calculating Direct Material and Direct Labor Variances [LO 9-3 Betty's Bakery has the following standard cost sheet for one unit of its most popular cake: SQ SP Direct materials 1.2...
-
eBook Replacement Analysis Although the Chen Company's milling machine is old, it is still in relatively good working order and would last for another 10 years. It is inefficient compared to modern...

Study smarter with the SolutionInn App


