What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3
Question:
What kind of hybridization do you expect for each carbon atom in the followingmolecules?
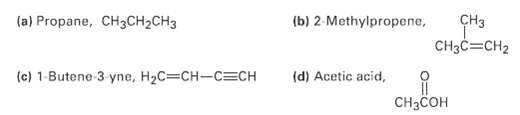
Transcribed Image Text:
(a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CHз CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHзCсон
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (18 reviews)
a sp3 sp3 sp3 CH3C...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H. C-H (a) 1 "CH-CH CH2 C=C H2N H. Procaine Vitamin C (ascorbic acid)
-
What hybridization do you expect for the atom indicated in red in each of the following species? (a) CH3CO2- (b) PH4+ (c) AlF3 (d) H2C==CH-CH2+
-
What dividends do you expect for Goodman Industries stock over the next 3 years if you expect the dividend to grow at the rate of 5% per year for the next 3 years? In other words, calculate D1, D2,...
-
Would individual mandates for health insurance be more or less burdensome to the poor than employer mandates? Would lower-income groups be wise to favor one plan over the other?
-
Art Funkel started his incorporated medical practice on June 1, 2016. He immediately made an Selection for the corporation. Art would like the corporation to adopt a tax year ending May 31 so that a...
-
a. From a process standpoint, evaluate how well this investigation began to unfold. b. Should Barbara have done anything differently? c. What changes, if any, would you recommend be made to the...
-
Atlanta Corp. is preparing its 2020 financial statements (December 31 year-end) and is considering the following events: a. Probable warranty costs on the company's products are estimated to be \(1...
-
Connor Corporation sells rock-climbing products and also operates an indoor climbing facility for climbing enthusiasts. During the last part of 2012, Connor had the following transactions related to...
-
Please help me find the character of the gain or loss as well as the basis in the stock. 6. Corporate formation (20 points) In 2020, Mark, Alice, Howard, Frances, and Vern formed Mel's Diner Inc....
-
Using Fig 2-14, find the daily evaporation from a shallow lake with the following characteristics Mean daily temperature = 25.6C, Daily solar radiation = 550 cal/cm2, Mean daily dew point = 4.4C,...
-
Convert the following molecular formulas into line-bond structures that are sonsistent with valence rules: (a) C3H8 (b) CH5N (c) C2H6O (2 possibilities) (d) C3H7Br (2 possibilities) (e) C2H4O (3...
-
What is the shape of benzene, and what hydribization do you expect for eachcarbon? C=c H-C - Benzene C-C I.
-
The drawing shows a wire tooth brace used by orthodontists. The topmost tooth is protruding slightly, and the tension in the wire exerts two forces T(vector) and T(vector) on this tooth in order to...
-
use logarithmic differentiation to find the derivative of the function: y = x x - 9 2
-
0.2 kg/s of alcohol is to be cooled from 75C to 35C in a counter flow heat exchanger. Cooling water enters the exchanger at 12C and at the rate of 0.16 kg/s. The convection heat transfer coefficient...
-
what ways do hierarchical organizational structures influence decision-making efficiency and employee autonomy, and how might these factors affect innovation and adaptability in a rapidly changing...
-
Customer Relationship Marketing (CRM) calls for a robust approach that eclipses a 360 view of the customer through development, growth and maintenance of long term cost effective relationships with...
-
How do social movements mobilize collective action and challenge dominant power structures to effect social change and promote social justice ?
-
For each case in Table 3.2, for the governing equation \(\ddot{x}+2 \zeta \omega_{n} \dot{x}+\omega_{n}^{2} x=A \cos \omega t\), evaluate \(C\) and \(\phi\), and \(B_{1}\) and \(B_{2}\) in Equation...
-
Presented below are income statements prepared on a LIFO and FIFO basis for Kenseth Company, which started operations on January 1, 2024. The company presently uses the LIFO method of pricing its...
-
Calculate the vapor pressure of n-butane as a function of temperature using the Peng-Robinson equation of state. Compare your results with (a) Literature values (b) Predictions using the...
-
Explain whether each pair of models represents isomers or the same compound. (All represent compounds with the formula C7H16.) Draw structures for each compound represented by the models.
-
Explain whether each pair of models represent isomers or the same compound. Draw structures for each compound represented by the models.
-
The following models represent three isomers of C6H4Cl2. Explain which of these compounds does not have a dipole moment.
-
Under the economies of scale principle of transportation, the cost per unit increases as the size of shipment increases. True False
-
Which of the alternatives has the lowest standard deviation? A. 1 B. 2 C. 3 D. All three alternatives have the same standard deviation.
-
Online presentations require complete familiarity with digital communication tools. True False

Study smarter with the SolutionInn App


