What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H.
Question:
What kind of hydribization do you expect for each carbon atom in the followingmolecules?
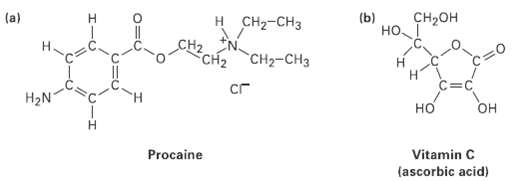
CH-он (b) H. н Cна-сHз (a) но 1 "CH-CHз CH2 C=C сг H2N H. но он Procaine Vitamin C (ascorbic acid)
Step by Step Answer:
a HN St H 302 H sp3 sp CC2 H ...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Organic Chemistry questions
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
What dividends do you expect for Goodman Industries stock over the next 3 years if you expect the dividend to grow at the rate of 5% per year for the next 3 years? In other words, calculate D1, D2,...
-
What hybridization do you expect for the atom indicated in red in each of the following species? (a) CH3CO2- (b) PH4+ (c) AlF3 (d) H2C==CH-CH2+
-
Discuss the ways that managed care organizations can infl uence the adoption of new technologies.
-
Review Examples 4 and 6 in the text. In both examples, the taxpayer's AGI is $129,400 even though in Example 6 there is $700 of nonrecaptured 1231 loss from 2015. Explain why the two AGI amounts are...
-
How should an expert witness deal with opposing experts?
-
Bloy Company pays all salaried employees on a biweekly basis. Overtime pay, however, is paid in the following biweekly period. Bloy accrues salaries expense only at its December 31 year-end. Data...
-
PC-Express is a computer retail store that sells two kinds of microcomputers: desktops and laptops. The company earns $600 on each desktop computer it sells and $900 on each laptop. The...
-
Question 1. IHI Take Test: Homework #8 ?? sum X @900 \ innmsaaa a; Question Completion Status: ! The management of the Diners Delight franchised restaurant chain is in the process of establishing...
-
Accentrics Limited has the following information available regarding its share capital at December 31, 2013: Preferred shares, $3.50 cumulative, 20,000 shares issued......................$1,000,000...
-
Potassium methoxide, KOCH3, contains both covalent and ionic bonds. Which do you think is which?
-
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions. Tell the hydribization, and predict the bond angles for each non-terminalatom. . Pyridoxal...
-
Portsmouth Company makes fine colonial reproduction furniture. Upholstered furniture is one of its major product lines and the bottleneck on this production line is time in the upholstery shop....
-
The following Income statement and additional year-end information is provided. SONAD COMPANY Income Statement For Year Ended December 31 Sales Cost of goods sold $ 1,599,000 783,510 Gross profit...
-
How does social stratification intersect with the dynamics of power and privilege in contemporary societies ?
-
Use the doubling time formula to estimate how long it would take a population of 5 billion to double given an annual growth rate of 1.5 percent.
-
Derive the conversion factors for the following quantities for volume flow rate a) Converting in/min to mm/s. b) Converting gallons per minute (gpm) to m/s. c) Converting gpm to liters/min. d)...
-
What is the relationship between innovation and organizational agility, and how can organizations encourage innovative thinking while maintaining operational effectiveness ?
-
A simple model of a seismometer is shown in Figure 3.60. The top view shows how a pen attached to the seismometer mass traces out a pattern on a paper grid that is on a roller. The side view provides...
-
Tell whether the angles or sides are corresponding angles, corresponding sides, or neither. AC and JK
-
Using the Redlich-Kwong equation of state, compute and plot (on separate graphs) the pressure and fugacity of nitrogen as a function of specific volume at the two temperatures a. 110 K b. 150 K
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Explain why one of these anions is much more stable than the other: : a) CH-C-CH-CH b) CH CH3 : CH-C-CH-CH CH-C=N:
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
1- Research has shown that shareholders of acquisition. a) acquiring firms b) target (acquired) firms c) both acquiring and target firms d) neither acquiring nor target firms earn above average...
-
Answer questions 1 & 2 based on the following scenario. (You should prepare a flexible budget to answer the questions) Door Draperies makes and sells curtains. Information related to its performance...
-
When companies reach the crossover point, they have filed for an initial public offering (IPO). their stock prices increase, because their earnings are now more predictable. their stock prices...

Study smarter with the SolutionInn App


