Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions.
Question:
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions. Tell the hydribization, and predict the bond angles for each non-terminalatom.
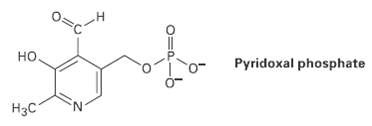
Transcribed Image Text:
но. Pyridoxal phosphate Нас 0=
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
sp3 sp2 H3C H sp2 sp2 sp2 sp2 5p3 Pyridoxa...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the molecular structure and the bond angles for each molecule or ion in Exercises 57, 58, and 60.
-
Predict the molecular structure and the bond angles for each of the following. a. SeO3 b. SeO2 c. PCl3 d. SCl2 e. SiF4
-
Predict the molecular structure and the bond angles for each of the following. (See Exercises 89 and 90.) a. XeCl2 b. ICl3 c. TeF4 d. PCl5
-
In the Akerlof example, the individuals are treated as indifferent to risk. What would you expect to see in these markets if individuals wanted to avoid risk? What if there were some risk lovers?
-
Glen and Diane Okumura (both age 48) are married, file a joint return, and live at 39 Kaloa Street, Honolulu, HI 96815. Glen's Social Security number is 111-11-1111, and Diane's is 123-45-6789. The...
-
For the three probability distributions shown, rank each distribution from lowest to highest in terms of the sample size required for the distribution of the sample mean to be approximately normally...
-
Stillsville Corporation manufactures an electronic switch for dishwashers. The cost base per unit, excluding selling and administrative expenses, is $60. The per unit cost of selling and...
-
A company is forecasting the purchase of inventory from an overseas vendor with payment to be made in a foreign currency (FC). Assume an option was used as a hedging instrument for this forecasted...
-
KHU, YU, and COHTLER operate under the name of KHUYUCOHT LTD. a partnership with a complex profit and loss sharing agreement. The average capital balance for each partner on December 31, 2020 is...
-
Use the data in the Excel file Baseball Attendance to do the following: a). Develop spreadsheet models for forecasting attendance using single moving average and single exponential smoothing. b)....
-
What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H. C-H (a) 1 "CH-CH CH2 C=C H2N H. Procaine Vitamin C (ascorbic acid)
-
Why do you suppose no one has ever been able to make cyclopentyne as a stable molecule? Cyclopentyne
-
Express f (x) + g(x) as a rational function. Carry out all multiplications. f(x) = x + 5 x - 10' g(x)= = x + 10
-
Dominique wanted a new building for her business. She obtained a bid of $10,000 to tear down her old building and another bid of $90,000 to replace it with a new one. Having only limited cash...
-
Sunland Company manufactures customized metal buildings. The following pertains to Job No. 953: Direct materials used $24800 Direct labor hours worked 600 Direct labor rate per hour $16 Machine hours...
-
Following the 2024 Kenya Certificate of Primary Education results, Azizi, Mlio, Haki and Dalila received admission to Great Kenya Secondary School. The school is one of the best institutions for...
-
An airplane flies 2 0 0 km due west from city A to city B and then 3 1 0 km in the direction of 3 1 . 5 \ deg north of west from city B to city C . ( a ) In straight - line distance, how far is city...
-
Critically analyse the major differences between traditional human resource management and strategic human resource management and assess the preposition that vertical fit is a prerequisite for...
-
What reasons might explain the (apparently) low usage rate of activitybased costing (ABC) systems in governmental organizations?
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
Can a fluid obeying the virial equation of state have a vapor-liquid transition?
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
If funds are withdrawn from an Oregon 529 College Savings Network plan for qualified education purposes, then which of the following is correct? Question 8Select one: a. The taxpayer will have an...
-
Nancy is single, 19 years old and a full-time college student in Oregon. Her parents claim her as a dependent. Nancy had earned and gross income of $820.00 for the year. Will Nancy be required to...
-
Use the australian accounting standard. On 1 July 2022, Surf Ltd leases a semi-trailer truck from Superior Ltd. The truck will be used to move spare parts between Surf's various warehouses. The...

Study smarter with the SolutionInn App


