What is the shape of benzene, and what hydribization do you expect for eachcarbon? C=c H-C
Question:
What is the shape of benzene, and what hydribization do you expect for eachcarbon?
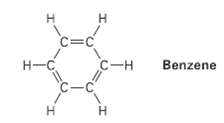
Transcribed Image Text:
C=c H-C С -н Benzene C-C I.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
HC CC 11 H CH 1 C 1 H Benzen...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the shape of the yield curve given in the following term structure? What expectations are investors likely to have about future interestrates? Term 1 year 2 years 3 years 5 years 7 years 10...
-
What is the shape of the chi-square distribution?
-
What is the shape of a monopolists demand curve and marginal revenue curve?
-
Distinguish between a National Health Insurance system and a National Health Service. Provide examples of each. What kind of a system does the United States have?
-
Shondee Corporation uses the lower of cost or market and FIFO inventory methods. At the end of 2015, the FIFO cost of the ending inventory was $181,000, and the market value of the inventory was...
-
A manager is suspected of making top-side journal entries to increase the income of the company because his bonus depends on the companys achieving at least a certain level of income. What would you,...
-
Stony Electronics, an electronics manufacturer, includes battery packs with its laptop computers that it sells. After a year of sales of the product, management learns that the battery packs have the...
-
On January 1, 2015, the ledger of Accardo Company contains the following liability accounts. Accounts Payable ......... $52,000 Sales Taxes Payable ........ 7,700 Unearned Service Revenue .........
-
THIS IS AN ACCOUNTING QUESTION. QUESTION 5 A. Case lmpian Company, a trading company needs to prepare a budget for April 2016. The following is information related to the company's plans for 2016: 1....
-
At the beginning of June 2021, Willingham Distributing Companys ledger showed Cash $18,000, Merchandise Inventory $5,900, and D. Willingham, Capital $23,900. During the month of June, the company had...
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
What bonds angles do you expect for each of the following, and what kind of hydribization do you expect for the central atom ineach? (a) (b) H. OH O (c) H2N-CH2-C-OH CHCH Lactic acid (in sour milk)...
-
Which characteristics of life can you identify in yourself ?
-
Thalassines Kataskeves, S.A., of Greece makes marine equipment. The company has been experiencing losses on its bilge pump product line for several years. The most recent quarterly contribution...
-
What are the implications of postcolonial theory for understanding the legacies of colonialism and ongoing processes of decolonization in globalized societies ?
-
2. For the given structure and boundary conditions, find the allowable load Pallow given the material strengths are: Tf=210 MPa for pins C and D in shear; and of=400 MPa for bars BC and DE in...
-
Discuss an outline of your SWOT analysis identifying the organizational competencies and strategic advantages. strengths include a strong popularity in healthcare, a strong group of employees, strong...
-
DEFINE THE TERM AUDIT AND EXPLAINING THE POINTS THAT AN AUDITOR HAS TO SATISFY HIMSELF BEFORE EXPRESSION ON OPINION.
-
Derive Equations 3.44 and 3.45: \[ \begin{aligned} |G(i \omega)| & =\frac{1}{k \sqrt{\left(1-\left(\omega / \omega_{n} ight)^{2} ight)^{2}+\left(2 \zeta \omega / \omega_{n} ight)^{2}}} \\ \phi &...
-
Diamond Walker sells homemade knit scarves for $25 each at local craft shows. Her contribution margin ratio is 60%. Currently, the craft show entrance fees cost Diamond $1,500 per year. The craft...
-
Proteins can exist in one of two states, the active, folded state and the inactive, unfolded state. Protein folding is sometimes thought of as a first-order phase transition from folded to unfolded...
-
Draw the structures of the compounds represented by these models and explain which would have the larger solubility in water. (The red atoms are oxygens.)
-
Draw the structures of the compounds represented by these models and explain which would have the higher melting point.
-
These models show the three isomers of C5H12. Explain which isomer would produce only one C5H11Cl isomer on reaction with Cl2 in the presence of light.
-
What happens to the price of both premium bonds and discount bonds as time progresses? They both decline in value They both converge on par as maturity approaches They both have unpredictable price...
-
When a preferred stock is "participating", it means that: A.Preferred stockholders may be entitled to payments above the normal level of dividends in certain situations. B.The prices of preferred...
-
1.To which input is mean-variance optimization most sensitive? a.expected variance b.historical correlation c.expected mean return

Study smarter with the SolutionInn App


