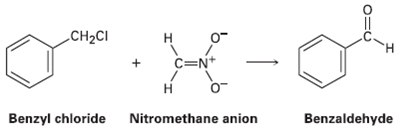
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves
Question:
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves initial conversion of nitro methane into its anion, followed by SN2 reaction of the anion with benzyl chloride and subsequent E2 reaction. Write the mechanism in detail, using curved arrows to indicate the electron flow in each step.
Transcribed Image Text:
CH2CI н C=N+ Н Benzyl chloride Nitromethane anion Benzaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
09100 CH 0 ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using curved arrows to indicate the election flow in each step, show how the base-catalyzed reverse aldol reaction of 4-hydroxy-4-methly-2-pentanone takes place to yield 2 equivalents of acetone.
-
Benzyl bromide is converted into benzaldehyde by heating in dimethyl sulfoxide. Propose a structure for the intermediate, and show the mechanisms of the two steps in thereaction. CH2BR E2 reaction...
-
Show how 4-methylcyclohexyl chloride can be converted to 4- methylcyclohexanecarboxylic acid.
-
IFRS requires the following: (a) Ending inventory is written up and down to net realizable value each reporting period. (b) Ending inventory is written down to net realizable value but cannot be...
-
The model below depicts industry analysis as a funnel that focuses on remote-factor analysis to better understand the impact of factors in the operating environment. Do you find this model...
-
The pie chart is drawn correctly, but it gives an incorrect impression. a. The area of each circle segment should be proportional to the percentage it represents. Explain how you can use the ribs of...
-
For each of the following, indicate whether the item is an adjustment to the bank balance or the book balance: 1. Bank service charge 2. Deposit in transit 3. Bank collection of amount due from...
-
Performance-Based Share Option Plan On January' 1, 2016, Rhine Company adopts a performance-based share option plan for its 80 key executives. Each executive is granted a maximum of 70 share options,...
-
On 10 May an investor sells a six-month put option on 1,000 shares of a stock. The current stock price $39 and the exercise price of the put option is $40. The price of the put option is $2.30 per...
-
Jenny Brown opened a law office, on July 1, 2010. On July 31, the balance sheet showed Cash $5,000, Accounts Receivable $1,500, Supplies $500, Office Equipment $6,000, Accounts Payable $4,200, and...
-
Rank the following substituted phenols in order of increasing acidity, and explain your answer: CH
-
Reduction of 2-butanone with NaBH4 yields 2-butanol. Is the product chiral is it optically active? Explain.
-
Give the expected major product(s) of each of the following electrophilic substitution reactions. N(CH3)2 CI CCH,CH, CH.CCI, AICI, Br,, FeBr, HNO,. H,SO, (a) (b) H;C CH; CH CH;0 NO2 SO,, H,SO, CISO,H...
-
What's your opinion on the Mc Donald's theory of world peace? What do you feel the most difficult part of international marketing is?
-
Choose the new media category. The new media is revolutionizing this generation we live in. I chose new media because it helps spread information and news virally and efficiently. Prompt Through the...
-
Aquamarine Company has the following information for the month of May of the current year: Selling price $220 per unit Variable production costs $90 per unit produced Variable selling and admin....
-
SoftwareExperts Inc produces an innovative and popular finance software. The company hires the most qualified programmers with an annual salary of 120,000$ to work on the project. Each programmer...
-
Depending on the profit margin of business. If I am looking to increase profit, then I would consider doing business with them. 25% seems like great profit margins, so I would at least place one...
-
a. Begin with one population and assume that \(y_{1}, \ldots, y_{n}\) is an i.i.d. sample from a Bernoulli distribution with mean \(\pi\). Show that the maximum likelihood estimator of \(\pi\) is...
-
Refer to the information from Exercise 22-19. Use the information to determine the (1) Weighted average contribution margin , (2) Break-even point in units, and (3) Number of units of each product...
-
The enthalpy of formation of NaI(s) is -288 kJ mol -1 . Use this value, together with other data in the text, to calculate the lattice energy of NaI(s).
-
List the following compounds in order of decreasing reactivity toward CH3O- in an SN2 reaction carried out in CH3OH: CH3F, CH3Cl, CH3Br, CH3I, CH3OSO2CF3, 14CH3OH.
-
Starting with (S)-2-bromobutane, outline syntheses of each of the following compounds: (a) (b) (c) (d) (R)-CH3CHCH2CH3 OCH2CH3 (R)-CH3CHCH2CH3 CCH3 (R)-CH3CHCH2CH3 SH (R)-CH3CHCH2CH3 SCH3
-
Which alkyl halide would you expect to react more rapidly by an SN2 mechanism? Explain your answer. (a) (b) (c) (d) (e) Br r or or CI
-
Must be in context to labor and employee relation and not in general Five Key Cases [Wm. Scott, Steel Equip., Renaud, Meiorin, Fraser Lakes] that LR pros refer to (noting what they are used for...
-
Body Images Pty Ltd (BI) is a beauty therapy company in Perth, Western Australia. They sell various beauty services to clients who want to improve their personal image. BI operates out of a square...
-
What sets Yorkville University apart from other institutions in terms of program offerings? How does Yorkville University's commitment to personalized learning enhance the student experience? What...

Study smarter with the SolutionInn App


