Convert these skeletal structures to line structures: OCH, CI b) a) d) - c)
Question:
Convert these skeletal structures to line structures:
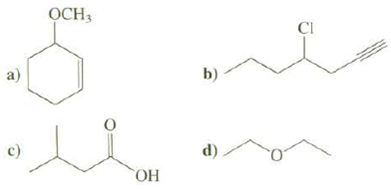
Transcribed Image Text:
OCH, CI b) a) d) - c) ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a HC H H 0CH C HI C ...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Convert the following condensed structures into skeletal structures (remember that condensed structures show atoms but few, if any, bonds, whereas skeletal structures show bonds but few, if any,...
-
Convert these structures to skeletal structures: T a) H-C-C H H H-C-H C c) CH3CHCCHCH(CH3)2 C=C=C-H b) H H-C-H H H CH3 d) CH,CHCH,NHCH,CH CH,
-
Convert the following structures into skeletal drawings: (a) (b) H. H. H. Indole 1,3-Pentadiene (d) (c) H. -CI C-CI H. 1,2-Dichlorocyclopentane Benzoquinone U
-
In a 1994 study, 164 pregnant, HIV-positive women were randomly assigned to receive the drug AZT during pregnancy and 160 such women were randomly assigned to a control group that received a placebo....
-
Describe the advantages and limitations of the four types of benchmarking?
-
Lei Materials balance sheet lists total assets of $1 billion, $100 million in current liabilities, $400 million in long-term debt, $500 million in common equity, and 50 million shares of common...
-
Interest rate swaps can be designated as hedges of underlying borrowings. A swap of floating for fixed rate is a cash flow hedge, but a swap of fixed for floating is a fair value hedge. Why?
-
Procter & Gamble has been the leading soap manufacturer in the United States since 1879, when it introduced Ivory soap. However, late in 1991, its major rival, Lever Bros. (Unilever), overtook it by...
-
3x-6y+z = 5 -x+y z=-1 x-2y= 2 by converting to a matrix equation and using the inverse of the coefficient matrix. Solve the system of equations 8
-
Icebreaker Company (a U.S.-based company) purchases materials from a foreign supplier on December 1, 2020, with payment of 22,000 dinars to be made on March 1, 2021. The materials are consumed...
-
Calculate the DU for these formulas and draw two constitutional isomer for each: (a) C 10 H 22 (b) C 9 H 16 (c) C 6 H 6
-
Determine whether these structures represent the same compound or isomers: ext0Q L a) O b) d) CH3 e) CH3CHCHCHCH3 CHCHCHCHCH 3 CH, f) CHCHCHCHCHCH3 CHCHCHCHCHCH, CH3 CH3 ~
-
In Table 11.1 in Chapter 11 we saw data on the state means of students who took the SAT exam. The mean Verbal SAT for North Dakota was 515. The standard deviation was not reported. Assume that 238...
-
BU Hotel Inc. has a beta of 2.6 and SHA Hotel Inc. has a beta of 0.90. The return on the market is 10% and the risk-free rate is 3%. If you wish to hold a portfolio consisting of BU and SHA Hotel and...
-
Green Company constructed a machine at a total cost of $73.90 million. Construction was completed at the end of 2020 and the machine was placed in service at the beginning of 2021. The machine was...
-
A (1.50x10^1) liter bottle is filled with nitrogen (N2) at STP and closed tight. (STP means "Standard Temperature and Pressure", i.e. 273 K and 1 atm.). To three significant figures, how many...
-
ABC Registered, a sole proprietorship, began carrying on a business on September 1, 2023. A calendar based fiscal period ending December 31 was chosen. On October 1, 2023 the proprietorship purchased...
-
Mary carries on business as a sole proprietor that generated $100,000 in net accounting income. Included in this amount are: $7,000 of amortization expense; $4,000 for bad debt expense; $112,000 cost...
-
Answer the following questions. a. What is the purpose of the second and third general attestation standards shown in Figure 2.8? b. In Figure 2.9, why is there no attestation standard of fieldwork...
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
The graph of a function f is shown in Figure 6. (a) Find the values of f(1) and f(5). (b) What are the domain and range of f? Figure 6 y 0 x
-
Aromatic iodination can be carried out with a number of reagents, including iodine mono chloride, ICI. What is the direction of polarization of ICI? Propose a mechanism for the iodination of an...
-
The sulfonation of an aromatic ring with SO3 and H2SO4 is reversible. That is, heating benzenesulfonic add with H2SO4 yields benzene. Show the mechanism of the de-sulfonation reaction. What is the...
-
The carbocation electrophile in a FriedelCrafts reaction can be generated in ways other than by reaction of an alkyl chloride with A1Cl3. For example, reaction of benzene with 2-methylpropene in the...
-
In evaluating the financial performance of a business that is for sale, the potential new owner should compare financial ratios to: Multiple Choice their business plan. their largest competitor....
-
The following day you receive a $600 cash payment for a two week (14 days) rental period, starting the 8th of February 2022. This payment is received from Paul Stone, 2 Watchtower Way, Torquay. There...
-
Gastow Pumps is a manufacturer of commercial and heavy industrial Pumps. The firm's two product lines are called Directlift and Gravity. The primary raw materials are flexible steel sheets, and 23cm...

Study smarter with the SolutionInn App


