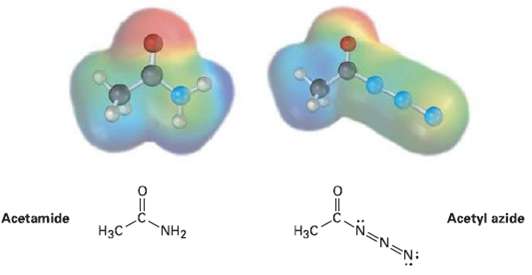
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which
Question:
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more reactive in nucleophilic acyl substitution reactions? Explain.
Transcribed Image Text:
Acetamide Acetyl azide NH2 Нзс Нас
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
According to the electrostatic potential maps the carbonyl carbon of ac...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which store do you think is more expensive-physical or online? A recent survey (USA Today, December 10, 2012, p. 1B) found that 46% of people aged 20 to 40 thought that physical stores were more...
-
Which of the alkyl halides is more reactive in an E2 reaction? a. CH3CH2CH2Br or b. c. d. CH,CH2CHCH3 Cl or CH cHy CH3CHCH2CHCH or CH3CH2CH2CCH3 Br Br CH, CH CH3 CH3
-
Which company and business model do you think is most likely to dominate the Internet and why ?
-
Give the approximate temperature at which creep deformation becomes an important consideration for each of the following metals: nickel, copper, iron, tungsten, lead, and aluminum.
-
What are detractors saying about PowerPoint, and why are they condemning it? Can you present a counterargument?
-
Describe and evaluate the use of return on investment (ROI) and economic value added (EVA) as growth targets in financial planning. Why do firms often use annual growth in sales or assets as a target...
-
What is a prospectus used for?
-
A trader wishes to unwind a position of 200,000 units in an asset over eight days. The dollar bidoffer spread, as a function of daily trading volume q, is a + b cq where a = 0.2, b = 0.15 and c = 0.1...
-
YouTube Maps w Extra Credit Peds W5 Flashca Med Admin Query. Results Gd pate-Doog Stream TV and M Help Save & Exit Submit Check my work Required information (The following information applies to the...
-
Rahim, a first-year auditor, is asked to select a sample of invoices to audit the utility expense account. Below is the account detail. The audit program asks to select a sample of four items. Month...
-
The following structure represents a tetrahedral alkoxide-ion intermediate formed by addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the...
-
Give IUPAC names for the following compounds: o (c) CHH2H2 CH2CH3 (a) (b) CH3CH2CHCHI NH2 (d) (e) (f) CH3CHCH,NHCH3 CH-CH-C H CH Br (h) (g) SCH(CH3)2
-
Provide some examples of front-of-the-house and back-of-the-house service operations.
-
80-kg person sits 3.0 m from the center of a horizontally rotating platform with a tangential speed of 2.0 m/s. What is the angular momentum of the person?
-
A stock had returns of 14.35 percent, 18.75 percent, -14.55 percent, 12.35 percent, and 25.03 percent for the past five years. What is the variance of the returns?
-
Your father is 50 years old and will retire in 10 years. He expects to live for 25 years after he retires, until he is 85. He wants a fixed retirement income that has the same purchasing power at the...
-
Annabelle generates revenue by selling toy for $5. The variable cost of goods sold per toy is $1, with a selling commission of 10 percent of sales. Fixed manufacturing costs total $25,000 per month,...
-
What advanced seismic retrofitting techniques are being employed to increase the resilience of existing infrastructure, and how do they compare to traditional methods in terms of cost and...
-
Dennis Kozlowski was a dominant, largerthan- life CEO of Tyco International, Ltd, a multi-billion-dollar company whose shares are still traded on the New York Stock Exchange (Symbol: TYC). His...
-
Accounting policies and practices that are most important to the portrayal of the companys financial condition and results, and require managements most difficult, subjective, or complex judgments...
-
Write orbital diagrams (boxes with arrows in them) to represent the electron configuration of carbon before and after sp 3 hybridization.
-
Give the structure that corresponds to each of the following molecular formulas and NMR spectra: (a) C5H10; 1.5, s (b) C2H2F3I: 3.56 (q, J = 10 Hz) (c) C6H14O: 0.91 (6H, d, 7 = 7 Hz); 1.17(6H,...
-
A compound A reacts with H2 over Pd/C to give methylcyclohexane. A colleague, A1 Keen, has deduced that the compound must be either 1-methylcyclohexene or 3-methylcyelohexene. You have been called in...
-
A compound A reacts with H2 over Pd/C to give methylcyclohexane. A colleague, A1 Keen, has deduced that the compound must be either 1-methylcyclohexene or 3-methylcyelohexene. You have been called in...
-
Blossom Fiber Company is the creator of Y-Go, a technology that weaves silver into its fabrics to kill bacteria and odor on clothing while managing heat. Y-Go has become very popular in undergarments...
-
Supongamos que el estado de flujos de caja de una empresa muestra un aumento de la tesorera. Cul de las siguientes transacciones podra contribuir al aumento de efectivo? Una disminucin de las cuentas...
-
Required information [The following information applies to the questions displayed below.] In each of the cases below, assume Division X has a product that can be sold either to outside customers or...

Study smarter with the SolutionInn App


