Give IUPAC names for the following compounds: o (c) CHH2H2 CH2CH3 (a) (b) CH3CH2CHCHI NH2
Question:
Give IUPAC names for the following compounds:
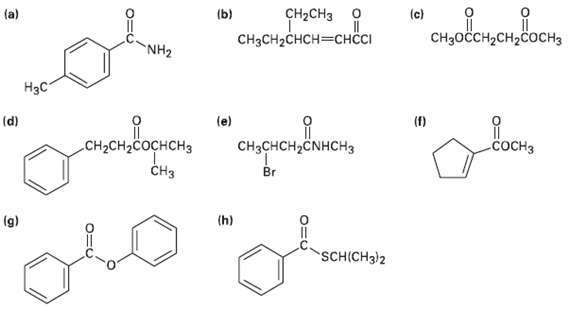
Transcribed Image Text:
oн (c) CHзоссH2сH2соснз CH2CH3 о (a) (b) CH3CH2CHCH—снссI NH2 Нзс (d) (e) (f) CH3CHCH,ČNHCH3 CH-CH-Cоснснз сосHз CHз Br (h) (g) SCH(CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (22 reviews)
a d H3C pMethylbenzamide g NH Isopropyl 3phenylpropanoate b CH3...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following compounds: CH CH3CH2CH=CCH2CH3 (b) (a) CH CHCH-H CHCH2CH2CHCHCHCH2CH3 CH CH (d) (c) CHCHCHCHC CH
-
Give IUPAC names for the followingcompounds: H CHCH2CCH H CH CHH2CH2CH (b) (e) CHH2H2C CH (a) H-H H CH3CH2CHCH2CH,CHCH3 (e) CH H CHCH2CH2CHCH2CH CH CH-H>H3 (d)
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Compare sand, die, investment, lost foam, and continuous casting techniques.
-
"Communicate-don't decorate." This principle is one of 20 rules that graphic designer and educator Timothy Samara discusses in his book Design Elements: A Graphic Style Manual. How could you apply...
-
What is the logic of the percentage-of-sales method for constructing pro forma statements?
-
Company X has capital of 2million shares that are currently trading at BC2000 per share.On its balance sheet it has a liability for an issue of convertible bonds with the following characteristics: ...
-
Boyne University offers an extensive continuing education program in many cities throughout the state. For the convenience of its faculty and administrative staff and to save costs, the university...
-
Part 4 of 8 Points: 0.19 of 4 The Everlasting Balloon Company produces party balloons that are sold in multi-pack cases. Following is the company's performance report in contribution margin format...
-
A group of your friends and you are very excited about setting up a business, in Indonesia selling imported home accessories and furniture sourced from all over the world. There are eight of you, who...
-
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more reactive in nucleophilic acyl substitution reactions?...
-
Draw structures corresponding to the following names: (a) p-Bromophenylacetamide (b) m-Benzoylbenzamide (c) 2, 2-Dimcthylhcxanamidc (d) Cyclohexyl cyclohexanecarboxylale (e) Ethyl...
-
Repeat Exercise 15.26 for water containing 0.324 mg/L of benzene (B) and 0.630 mg/L of m-xylene (X). Adsorption isotherms at 25C for these low concentrations are independent and given by The SOC...
-
The total value of cross-border trade a. is unknown. b. is calculated as a separate component of all international trade. c. is so small that countries do not bother to keep track of it. d. is...
-
Calculate the determinant of the following matrix by expansion along rows or columns of your choice. 1520 305T 0 0 -3 0 0 4 [0 -1 7 0
-
A student sits on a freely rotating stool holding two dumbbells, each of mass 3.09 kg (see figure below). When his arms are extended horizontally (Figure a), the dumbbells are 0.97 m from the axis of...
-
If x = 3y+4 then ex is equal to
-
Company XYZ is a leading technology firm known for its innovative products. They are planning to launch a new smartphone model in the market. As a design consultant, how would you approach the...
-
On March 19, 2003, the SEC filed accounting fraud charges in the Northern District of Alabama against HealthSouth Corporation and its CEO, Richard Scrushy. Scrushy was also charged with knowingly...
-
A company has the following incomplete production budget data for the first quarter: In the previous December, ending inventory was 200 units, which was the minimum required, at 10% of projected...
-
Write orbital diagrams (boxes with arrows in them) to represent the electron configurationswithout hybridizationfor all the atoms in SF 2 . Circle the electrons involved in bonding. Draw a...
-
How many absorptions should be observed in the 13C NMR spectrum of each of the following compounds? (Assume that the chair interconversion is rapid.) CH3 H,C
-
Explain how the proton NMR spectra of the compounds within each of the following sets would differ, if at all. (CH)2CHC and (CH CDCI
-
Although this chapter has discussed only nuclei that have spin 1/2, several common nuclei such as 14N and deuterium (2H, or D) have a spin of 1. This means that the spin has three equally probable...
-
Question: The Principle Limited Partnership has more than 300 partners and is publicly traded. The Principle was grandfathered under the 1987 Tax Act and has consistently been taxed as a partnership....
-
Wicker Corporation made estimated tax payments of $6000 in Year 1. On March 12 of Year 2, it filed its Year 1 tax return showing a $20,000 tax liability, and it paid the $14,000 balance at the time....
-
Assume you are wrapping up the 20X4 audit for your client, West Tech Inc. (WT), a manufacturer of cell phones, tablets, and personal computers. To date there has been every indication that the...

Study smarter with the SolutionInn App


