How would you synthesize the following substances starting frombenzene? ( C-CH2 .CH- (a) (c) H
Question:
How would you synthesize the following substances starting frombenzene?
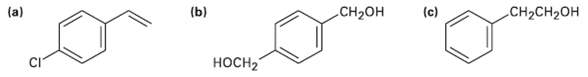
Transcribed Image Text:
(ы Cн-CH2он .CH-он (a) (c) носH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
All of these syntheses involve NBS bromination of the benzylic pos...View the full answer

Answered By

Milan Mondal
I am milan mondal have done my Msc in physics (special astrophysics and relativity) from the University of burdwan and Bed in physical science from the same University.
From 2018 I am working as pgt physics teacher in kendriya vidyalaya no2 kharagpur ,west bengal. And also I am doing advanced physics expert in chegg.com .also I teach Bsc physics .
I love to teach physics and acience.
If you give me a chance I will give my best to you.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you synthesize the following substances from benzaldehyde and any other reagentsneeded? (c) (b) (a) CH- CH2
-
How would you synthesize the following esters? a. N-octylacerate b. a404040404040-20H CH,CH2CH2CH2CH2CH2O-CCH2CH3
-
How would you synthesize the following compound starting from benzene? More than one step isneeded.
-
Jorge has investments in two limited partnerships, both of which constitute passive activities. This year, Jorges distributive share of income or loss from each partnership is as follows: In addition...
-
George is a U.S. citizen who is employed by Hawk Enterprises, a global company. Beginning on June 1, 2016, George began working in London. He worked there until January 31, 2017, when he transferred...
-
A 10%, 25-year bond has a par value of $1,000 and a call price of $1,075. (The bonds first call date is in 5 years.) Coupon payments are made semiannually. a. Find the current yield, YTM, and YTC on...
-
Lorch Company uses budgets in controlling costs. The May 2002 budget report for the company's Packaging Department is as follows. The budget amounts in the report were on the master budget For the...
-
McLain Corporation sold $6,000,000, 9%, 10-year bonds on January 1, 2014. The bonds were dated January 1, 2014 and pay interest on July 1 and January 1. McLain Corporation uses the straight-line...
-
Exercise 5-18 (Algo) Break-Even and Target Profit Analysis; Margin of Safety; CM Ratio [LO5-1, LO5-3, LO5-5, LO5-6, LO5-7] Menlo Company distributes a single product. The company's sales and expenses...
-
Colson Company has a line of credit with Federal Bank. Colson can borrow up to $800,000 at any time over the course of the Year 1 calendar year. The following table shows the prime rate expressed as...
-
As written, the following syntheses have flaws. What is wrong witheach? CH o (b) CI (a) .CH 1. Cl2, FeCl3 1. HNO3, H2SO4 2. CH3CI, AICI3 3. Fe, H30* 4. NaOH, H20 2. KMNO4 NH2 (c) CH3 CH NO2 1. CH3CI,...
-
The compound MON-0585 is a nontoxic, biodegradable larvicide that is highly selective against mosquito larvae. Synthesize MON-0585 using either benzene or phenol as a source of the aromaticrings....
-
An insulated cylinder fitted with a piston contains 0.1 kg of water at 100C, 90% quality. The piston is moved, compressing the water until it reaches a pressure of 1.2 MPa. How much work is required...
-
20. Enzymatic reactions can be expressed in which of the following chemical notations? a. S + P = E b. P + P = E c. E + S = P d. S + S = P 21. Enzymes that remove functional groups from a substrate...
-
Assume a company worked on four jobs during June: Jobs W , X , Y , and Z . At the end of June, the job cost sheets for these four jobs contained the following data: Job W Job X Job Y Job Z Beginning...
-
Joyce owns a gas station and monopolizes gas sales along a remote stretch of road. In February, Joyce stayed open even though she earned negative economic profits. a. Draw a correctly labeled graph...
-
Respond to what market considerations your classmates should consider for this species. What do you think about the possibility of introducing the species to the free market? What considerations...
-
1. Matrix formed outside the plasma membrane is referred to as a. cytoplasm. b. glycocalyx. c. exoplasm. d. cell wall. 2. The cell organelles found only in algae and plant cells are a. chloroplasts....
-
Eastern Company was organized when it acquired \($1,000\) cash from the issue of common stock. During its first accounting period the company earned \($800\) of cash revenue and incurred \($500\) of...
-
ABC company leased new advanced computer equipment to STU Ltd on 1 January 2019.STULtd has to pay annual rental of $290,000 starting at 1 January 2019. It is a four years lease with ultimate rental...
-
Isobutane is to be liquefied to make liquid petroleum gas (LPG). The butane is available at 25 C and 1 bar, it will be compressed to 15 bar, cooled to 0 C in a heat exchanger, and expanded and...
-
Show reactions that could be used to convert 1-penetene to these compounds. More than one step may bnecessary. b) CI d) . h) g)
-
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral. , CH,CH,CH3 a)...
-
This alkynes hydration reaction can occur without added Hg2+. Show all the steps in themechanism. H,SO, PhC=CH + H20 PHCCH3
-
Larry's Woodworks has collected the following data for its cutting board line of products: Direct materials standard Direct materials standard cost Actual Direct Materials Used (AQU) Actual finished...
-
DIF MOH HCM 416 Use the information for St. Vincent's Hospital. Assume the hospital uses salary dollars as the costs driver for general administration and employee benefits, and space utilization as...
-
Create an Excel spreadsheet to solve the following problems. 1. Daniel deposited a lump sum 10 years ago, earned 3% interest compounded monthly and now has $40,000. How much did he invest? a. The NBC...

Study smarter with the SolutionInn App


