Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write
Question:
Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write balanced equations for the reactions causing the solution to be acidic or basic. The relevant Ka and Kb values are found in Tables 13.2 and 13.3.
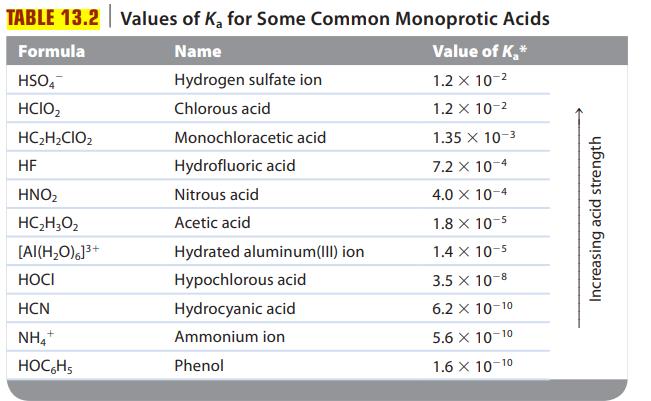
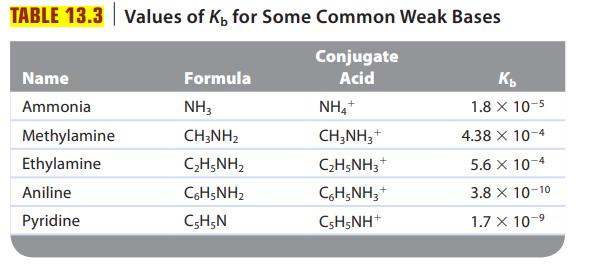
Transcribed Image Text:
TABLE 13.2 Values of K₂ for Some Common Monoprotic Acids Name Value of K₂* Hydrogen sulfate ion 1.2 x 10-² Chlorous acid 1.2 x 10-² 1.35 x 10-3 7.2 x 10-4 4.0 X 10-4 Formula HSO4 HCIO₂ HC₂H₂CIO₂ HF HNO₂ HC,H,Oz [AI(H₂O)]³+ HOCI HCN NH4+ HOCHS Monochloracetic acid Hydrofluoric acid Nitrous acid Acetic acid Hydrated aluminum(III) ion Hypochlorous acid Hydrocyanic acid Ammonium ion Phenol 1.8 x 10-5 1.4 x 10-5 3.5 x 10-8 6.2 X 10-10 5.6 X 10-10 1.6 X 10-10 Increasing acid strength
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Solutions of the following salts are acidic basic or neutral Salt Solution Equation NaHS Basic NaHS H2O HS Na H3O NH4Cl Acidic NH4Cl NH4 Cl NaCH3COO B...View the full answer

Answered By

Joemar Canciller
I teach mathematics to students because I love to share what I have in this field.
I also want to see the students to love math and be fearless in this field.
I've been tutoring these past 2 years and I would like to continue what I've been doing.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Lets take a look at the extensive career of Helen Frankenthaler (1928-2011), who found a way to incorporate all of the innovative techniques artists were experimenting with starting in 1945. Her...
-
Will 0.10 M solutions of the following salts be acidic, basic, or neutral? a. Ammonium bicarbonate b. Sodium dihydrogen phosphate c. Sodium hydrogen phosphate d. Ammonium dihydrogen phosphate e....
-
Decide whether solutions of the following salts are acidic, neutral, or basic. a. Ammonium acetate b. Anilinium acetate
-
Which of the following would be the most frequently occurring daily transaction in a retail shop? (a) Paying salary to the sales assistant (b) Sale of goods (c) Payment of rent for the shop premises...
-
James Lillards first wife had a child whom James adopted when he married that childs mother. James fathered other children with her until they divorced in the early 1970s. In 1975, James married his...
-
In the audit of Whirland Chemical Company, a large publicly traded company, you have been assigned the responsibility for obtaining background information for the audit. Your firm is auditing the...
-
Symbol \(C\) could represent (a) batched time cards. (b) unclaimed payroll checks. (c) erroneous time cards. (d) an error report.
-
Aquatic Biotechnology Inc. (ABI) is a medium-sized, public company operating an aquaculture business in eastern Canada. The company has been in operation since the mid-1990s, and during the latter...
-
Description Respond to the following scenario with your thoughts, ideas, and comments. Be substantive and clear, and use research to reinforce your ideas. Apix is considering coffee packaging as an...
-
An infinite cylinder of radius R carries a uniform surface charge density o. We propose to set it spinning about its axis, at a final angular velocity ws. How much work will this take, per unit...
-
Consider a solution of an unknown salt having the general formula BHCl, where B is one of the weak bases in Table 13.3. A 0.10-M solution of the unknown salt has a pH of 5.82. What is the actual...
-
The K b values for ammonia and methylamine are 1.8 10 -5 and 4.4 10 -4 , respectively. Which is the stronger acid, NH 4 + or CH 3 NH 3 + ?
-
Few corporate raiders are known for their integrity. Translate the following into standard-form categorical propositions.
-
Yuanne Sipp and Letitia Grimes share partnership income on a 3:2 basis. They have capital balances of $569,000 and $283,000, respectively, when Tammy Tuck is admitted to the partnership. Prepare the...
-
Sunlight at 0.64 m reflected by the Earth is absorbed by a nadir viewing satellite sensor at an altitude of 800 km. The sensor has a circular detector with a radius of 1 mm and a bandwidth of 0.1 m...
-
Determine the correct amount of net income for August and the total assets, liabilities, and owner's equity at August 31. In addition to indicating the corrected amounts, indicate the effect of each...
-
1. State the 4 major challenges or themes for IT leaders when it comes to managing IT innovation. 2. State some of the guiding principles for facilitating IT innovation. 3. What are some of the...
-
A straight 25 cm pipeline 5 km long is laid between two reservoirs having a difference of levels of 40 mtrs. To increase the capacity of the system, an additional 2.5 km long 2.5 cm pipe is laid...
-
The account balances of Wilford Towing Service at June 30, 2016, follow Equipment...........$ 17,500 Office Supplies ........... 1,300 Notes Payable........... 6,900 Rent Expense........... 800...
-
How can NAFTA be beneficial to suppliers of Walmart?
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
Lactones can be prepared from diethyl malonate and epoxides. Diethyl malonate is treated with a base, followed by an epoxide, followed by heating in aqueous acid: Using this process, identify what...
-
Predict the major product of the following transformation. CO2ET C10H100 Heat
-
The receipt of payment for an account receivable: affects assets and equity affects assets and liabilities affects only the asset side of the accounting equation has no effect on the accounting...
-
Dan got a 1 0 year Fixed Rate Mortgage for $ 1 0 0 , 0 0 0 . The loan has constant annual payments and an annual interest rate of 5 % . There are no closing costs. Suppose Dan prepays the loan in...
-
1) Why could quantum computing be a very important tool in supply chain management (what can it do that traditional computing can't)? 2) Give 1 or 2 examples of some current issues in supply chain...

Study smarter with the SolutionInn App


