Consider a solution of an unknown salt having the general formula BHCl, where B is one of
Question:
Consider a solution of an unknown salt having the general formula BHCl, where B is one of the weak bases in Table 13.3. A 0.10-M solution of the unknown salt has a pH of 5.82. What is the actual formula of the salt?
Table 13.3.
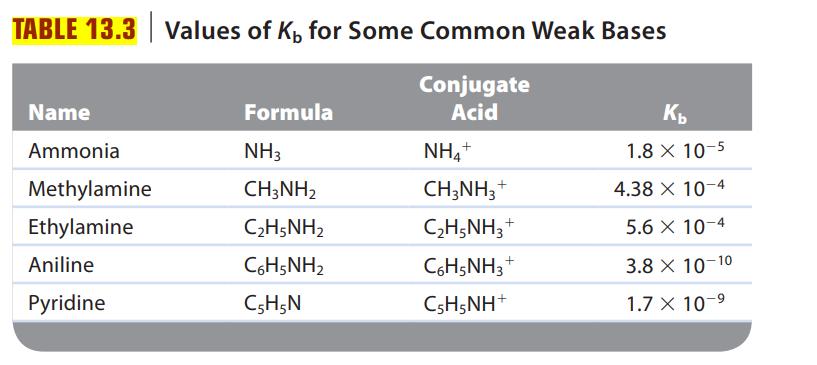
Transcribed Image Text:
TABLE 13.3 Values of K, for Some Common Weak Bases Conjugate Acid Name Ammonia Methylamine Ethylamine Aniline Pyridine Formula NH3 CH3NH₂ C2H5NH2 CoH5NH2 C5H5N NH4+ CH3NH3 + C₂H5NH3 C6H5NH3 CsH5NH* + + Kb 1.8 X 10-5 10-4 10-4 10-10 10-⁹ 4.38 x 5.6 x 3.8 X 1.7 X
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine the actual formula of the unknown salt we need to determine the identity of the weak ba...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
An aqueous solution of an unknown salt of gold is electrolyzed by a current of 2.75 amps for 3.50 hours. The electroplating is carried out with an efficiency of 90.0%, resulting in a deposit of...
-
An aqueous solution of an unknown salt of vanadium is electrolyzed by a current of 2.50 amps for 1.90 hours. The electroplating is carried out with an efficiency of 95.0%, resulting in a deposit of...
-
Separate samples of a solution of an unknown salt are treated with dilute solutions of HBr, H2SO4, and NaOH. A precipitate forms in all three cases. Which of the following cations could the solution...
-
If net assets of a business totalled 200,000 and its total assets on that date amounted to 325,000, its liabilities would amount to: (a) 125,000 (b) 200,000 (c) 525,000 (d) Not sufficient information...
-
Vickie Lynn Smith, an actress and model also known as Anna Nicole Smith, met J. Howard Marshall II in 1991. During their courtship, J. Howard lavished gifts and large sums of money on Anna Nicole,...
-
Statements of earnings and financial position for Prairie Stores Corporation are shown on the next page. REQUIRED a. Use professional judgment in determining materiality based on revenue, net income...
-
Which of the symbolic representations in Figure 2-24 indicates that a sales invoice has been filed? Figure 2.24 [a] [b] [c] [p]
-
Birmingham Bowling Ball Company (BBBC) uses a job-order costing system to accumulate manufacturing costs. The companys work-in-process on December 31, 20x3, consisted of one job (no. 3088), which was...
-
Find the length of the curve. x = 10t, y = 15t, 0sts 3
-
By modern standards, the shaft design of Problem 5-76 is poor because it is so long. Suppose it is redesigned by halving the length dimensions. Using the same material and design factor as in Problem...
-
Calculate the pH of each of the following solutions. a. 0.12 M KNO 2 b. 0.45 M NaOCl c. 0.40 M NH 4 ClO 4
-
Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write balanced equations for the reactions causing the solution to be acidic or basic. The relevant Ka...
-
Presented here are the original overhead budget and the actual costs incurred during April for Piccolo Inc. Piccolos managers relate overhead to direct labor hours for planning, control, and product...
-
You and your significant other are planning to retire and open a chicken ranch in 40 years. However, you begin a retirement fund to make your dreams come true. You plan to invest $5,000 each year for...
-
As a long-term investment, Painters' Equipment Company purchased 20% of AMC Supplies Inc's 470,000 shares for $550,000 at the beginning of the fiscal year of both companies. On the purchase date, the...
-
The Freundlich isotherm is expressed as: W = k Po 1/n such that W is the loading, p is the pressure, po is the CO vapor pressure, and k and n are fitting parameters. Compare this fit to that of the...
-
A baseball of mass 0.3kg approaches the bat at a speed of 30 miles/hour, and when the ball hits the bat for 0.5 s, it starts to move away from the bat at a speed of 60 miles/hour. Find the impulse?
-
Bob the Builder and Wendy were partners in their constructionbusiness and were general contractors. They submitted a bid toconstruct a new building for the the city. They includedLumberjack\'s quote...
-
The account balances of Wilford Towing Service at June 30, 2016, follow Equipment...........$ 17,500 Office Supplies ........... 1,300 Notes Payable........... 6,900 Rent Expense........... 800...
-
How will relating product contribution margin s to the amount of the constrained resource they consume help a company maximize its profits?
-
Propose an efficient synthesis for the following transformation.
-
For a pair of keto-enol tautomers, explain how IR spectroscopy might be used to identify whether the equilibrium favors the ketone or the enol.
-
Acrolein is an α,β-unsaturated aldehyde that is used in the production of a variety of polymers. Acrolein can be prepared by treating glycerol with an acid catalyst. Propose...
-
Consider a situation where Ripple Blockchain Incorporated sells an asset after eight years of use, receiving $31,000 in cash. The asset was bought for $126,200 and was expected to last 10 years, with...
-
Suppose, risk-free rate is 1.5%. The optimal tangent portfolio is formed with 30% on Amazon and 70% on Pfizer. The optimal risky portfolio has expected return of 24%, standard deviaiton of 15%, and...
-
Step 2: Based on the findings from your research, decide on three potential price points for BOTH your 500g and 1kg honey products and run a break-even analysis for each price point. For the purpose...

Study smarter with the SolutionInn App


