Assign oxidation numbers to all the atoms in each of the following: a. HNO3 b. CuCl c.
Question:
Assign oxidation numbers to all the atoms in each of the following:
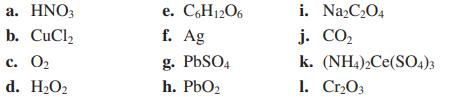
Transcribed Image Text:
a. HNO3 b. CuCl₂ c. 0₂ d. H₂O₂ e. C6H12O6 f. Ag g. PbSO4 h. PbO₂ i. Na₂C₂O4 j. CO₂ k. (NH4)2Ce(SO4)3 1. Cr₂03
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The oxidation numbers of all the atoms in each of the following compounds are a HNO3 H1 N5 O2 b CuCl ...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Assign oxidation numbers to all the atoms in each of the following: a. HNO 3 b. CuCl 2 c. O 2 d. H 2 O 2 e. C 6 H 12 O 6 f. Ag g. PbSO 4 h. PbO 2 i. Na 2 C 2 O 4 j. CO 2 k. (NH 4 ) 2 Ce(SO 4 ) 3 l....
-
Assign oxidation numbers to the atoms in each substance. a. Kr (krypton) b. krypton tetrafluoride (KrF4) c. dioxygen difluoride (O2F2)
-
Assign oxidation numbers to the atoms in each substance. a. Lithium hydride (LiH) b. Potassium peroxide (K2O2) c. Potassium fluoride (KF)
-
I recently heard my neighbor discussing how one of our other neighbors lost his job. My neighbor assumed that the person who lost his job was probably lazy or not smart enough for the job, without...
-
Prepare the journal entries to record these transactions on Kesler Companys books using a periodic inventory system. (a) On March 2, Kesler Company purchased $800,000 of merchandise from Rice...
-
Gordon Driving Schools 2014 balance sheet showed net fixed assets of $1.65 million, and the 2015 balance sheet showed net fixed assets of $1.73 million. The companys 2015 statement of comprehensive...
-
The owner of a business introduced new capital of 15,000 during the year and withdrew a monthly salary of 2,000. If the net assets at 1 January 2008 and 31 December 2008 were 85,000 and 73,000...
-
Fill in the spreadsheet below and answer the following questions. Calculate answers to three decimal places. a. Calculate Total Returns (TRs) and Return Relatives (RRs) for McDonald's for the 10...
-
Listed below are the lead concentrations (in g/g) measured in different Ayurveda medicines. Ayurveda is a traditional medical system commonly used in India. The lead concentrations listed here are...
-
The two employees of Silver Co. receive various fringe benefits. Silver Co. provides vacation at the rate of $315 per day. Each employee earns one day of vacation per month worked. In addition,...
-
In the electrolysis of an aqueous solution of Na 2 SO 4 , what reactions occur at the anode and the cathode (assuming standard conditions)? 2- S0 +2e 250 O + 4H+ + 4e 2HO 2HO + 2e H + 2OH Na + e Na...
-
(a) How is resistance present in all transmission lines? (b) How is inductance present in all transmission lines? (c) How is capacitance present in all transmission lines? (d) The combined effects of...
-
Find an equation of the tangent plane to the given surface at the specified point. z = x sin(x + y), (1, 1, 0)
-
Adam Smith and John Maynard Keynes are two of the most influential economists in the history of the field. Give an example of a short essay comparing Smith's and Keynes's fundamental economic...
-
The industrial company Efficar is considering expanding its current product line by manufacturing a new product for which it would benefit from a growing market. This would require investment in new...
-
Two carts with masses of 4.80 kg and 3.42 kg move toward each other on a frictionless track with speeds of 5.39 m/s and 3.03 m/s respectively. The carts stick together after colliding head-on. Find...
-
A company must make a payment of $350,000 two years from today. How much must the company deposit per quarter in an account that earns 4% interest every quarter so that it has $350,000 at the end of...
-
Create a VIEW called customers_private_info that contains three columns: customer_id email_address password Create a trigger called customers_password_length_check that verifies the length of the...
-
In examining borrower characteristics versus loan delinquency, a bank has collected the following information: (1) 15% of the borrowers who have been employed at their present job for less than 3...
-
The activities listed in lines 2125 serve primarily as examples of A) Underappreciated dangers B) Intolerable risks C) Medical priorities D) Policy failures
-
Can a molecule with an inversion center have a dipole moment? Give an example of a molecule with this symmetry element and explain your reasoning.
-
Which of the three normal modes of H 2 O in Figure 27.9 is best described as a bending mode? Does the bond angle remain unchanged in any of the modes? Which requires less energy, bond bending or bond...
-
Why does the list of elements for the D 6h group in Table 27.2 not list the elements C 2 6 , C 3 6 , and C 4 6 ? Selected Point Groups and Their Elements Symmetry Elements TABLE 27.2 Example Molecule...
-
Question 20 (1 point) Listen The Carters contributed $8,000 to the Path for Progress Political Action Committee What is the tax treatment of this contribution? O Deductible on Schedule E as an...
-
The Sheffield and Frizell Dental Clinic provides both preventive and orthodontic dental services. The two owners, Reese Sheffield and Anita Frizell, operate the clinic as two separate investment...
-
Montoure Company utiliza un sistema de inventario peridico. Realiz las siguientes transacciones de compra y venta en el ao calendario. Fecha Actividades Unidades Adquiridas al Costo Unidades vendidas...

Study smarter with the SolutionInn App


