In the electrolysis of an aqueous solution of Na 2 SO 4 , what reactions occur at
Question:
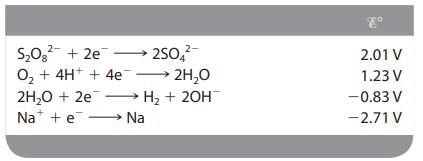
In the electrolysis of an aqueous solution of Na2SO4, what reactions occur at the anode and the cathode (assuming standard conditions)?
Transcribed Image Text:
2- S₂0² +2e 250 O₂ + 4H+ + 4e →→→ 2H₂O 2H₂O + 2e → H₂ + 2OH Na + e→→→→→→→→→ Na 2.01 V 1.23 V -0.83 V -2.71 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
In the electrolysis of an aqu...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write equations for the half-reactions that occur at the anode and cathode in the electrolysis of molten KBr. What are the products formed at the anode and cathode in the electrolysis of aqueous KBr?
-
Which product, O 2 or F 2 , is more likely to form at the anode in the electrolysis of an aqueous solution of KF? Explain your reasoning.
-
Predict the products formed in the electrolysis of an aqueous solution of CdSO 4 .
-
Using Figure 7-5 as an example, redraw Figure 7-12 using an enterprise information system that processes a shared database. Explain the advantages of this system over the paper-based system in Figure...
-
On October 5, Lane Company buys merchandise on account from OBrien Company. The selling price of the goods is $5,000, and the cost to OBrien Company is $3,000. On October 8, Lane returns defective...
-
In 2001, President George W. Bush and Federal Reserve Chairman Alan Greenspan were both concerned about a sluggish U.S. economy. They also were concerned about the large U.S. current account deficit....
-
A countercurrent leaching system is recovering oil from soybeans with five stages. On a volumetric basis, liquid flow rate/solids flow rate \(=1.36\). Recovery of oil in the solvent is \(97.5 \%\)....
-
What is a work breakdown analysis and why is this concept important?
-
For the following CLD develop a dynamic stock-flow diagram. (Include variable units) Hiring Hiring time Receiving Order Salesman Total Income Income Sales Efficiency Perceived Delay Correction Order...
-
Legendary for his business and investment acumen, Warren Buffett is frequently called the Oracle of Omaha after his birthplace in Omaha, Nebraska. As one of the worlds wealthiest business...
-
Gold is produced electrochemically from an aqueous solution of Au(CN) 2 - containing an excess of CN - . Gold metal and oxygen gas are produced at the electrodes. What amount (moles) of O 2 will be...
-
Assign oxidation numbers to all the atoms in each of the following: a. HNO3 b. CuCl c. 0 d. HO e. C6H12O6 f. Ag g. PbSO4 h. PbO i. NaCO4 j. CO k. (NH4)2Ce(SO4)3 1. Cr03
-
1. Describe the two different mental models represented in this story. 2. What are some of the assumptions that shape the mindset of Sam Nolan? Of Tom Carr? 3. Do you think it is possible for Carr to...
-
1) List and describe each of the seven interlinking functions of human resource management (HRM) b) Describe why HRM is critical to organizations Question 2) Why is it important for HR management to...
-
What are the one key organizational behavior factor contributing to the high turnover, and suggest a brief strategy to address it?
-
A soccer player kicks the ball with an initial velocity of 22.3 m/s [14 degrees above the horizontal] on a level playing field. Determine the soccer balls: a) total hang time b) maximum height...
-
1What will be the maturity value of a promissory note subscribed today for 150,000 at an interest rate of 27.5% if the term of the operation is 10 months? 2.What will be the maturity value of a...
-
On January 1, 2025, Sheridan Company had a balance of $410,400 of goodwill on its balance sheet that resulted from the purchase of a small business in a prior year. The goodwill had an indefinite...
-
A small British computer-game firm, Eidos Interactive PLC, stunned the U.S.- and Japan-dominated market for computer games when it introduced Lara Croft, an Indiana Jones-like adventuress. The...
-
Separate variables and use partial fractions to solve the initial value problems in Problems 18. Use either the exact solution or a computer-generated slope field to sketch the graphs of several...
-
A surface for which the electrostatic potential is negative delineates regions in a molecule that are subject to electrophilic attack. It can help you to rationalize the widely different chemistry of...
-
Hydrocarbons are generally considered to be nonpolar or weakly polar at best, characterized by dipole moments that are typically only a few tenths of a debye. For comparison, dipole moments for...
-
Chemists know that nitric and sulfuric acids are strong acids and that acetic acid is a weak acid. They would also agree that ethanol is at best a very weak acid. Acid strength is given directly by...
-
The following information is available for ABC Ltd Particulars Rs in million Net operating income(NOI) Rs 40 Interest on debt Rs 10 Cost of equity(Ke) 0.18 Cost of debt(Kd) 0.12 (a) What is the...
-
5. (a) Estimate the cost of preferred stock (required return of preferred stock). (b) Redo question 3 including preferred stock as a financing source, and assume the target weights are as follows:...
-
Suppose you are given the following data. You have run a three month moving average and found the forecasts, difference, and difference squared between the demand and forecasts. Note, the forecasts,...

Study smarter with the SolutionInn App


