In general, intramolecular reactions that form rings are often disfavored entropically because it makes a flexible starting
Question:
In general, intramolecular reactions that form rings are often disfavored entropically because it makes a flexible starting material more rigid. In the case of ring-closing metathesis, however, the entropy penalty for the closure is not especially large for most systems, such as the one below used as part of a synthesis of an anti-tumor compound known as epothilone A. Why might that be?
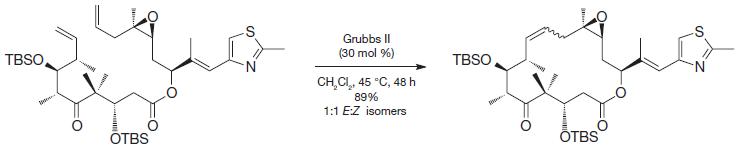
Transcribed Image Text:
TBSO THESE OTBS Grubbs II (30 mol %) CH,CI,, 45 °C, 48h 89% 1:1 E:Z isomers TBSO w OTBS N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
It is because of two factors 1 metathesis reactions proceed via a ringopening step which is much more acidic than a ring closing step and 2 the final product is a macrocycle which will be rigid the strain energy required to form a macrocycle is higher than that of an open chain molecule In addition one must also consider the driving force for the reaction G The G term overall comes from two components the enthalpy change of the metathesis reaction ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
A compound pendulum is defined as a rigid slab which oscillates about a fixed point O, called the center of suspension. Show that the period of oscillation of a compound pendulum is equal to the...
-
A spiro ring junction is one where two rings that share no bonds originate from a single carbon atom. Alkanes containing such a ring junction are called spiranes. (a) For the case of bicyclic...
-
Why should 3-methylcyclohexene not be used as the starting material in Problem 30b?
-
Create a T-Account Transaction Account titles Cash d Common stock Supplies Creditors accounts payable) Cash Fees earned Rent expense Cash Creditors(accounts payable) Cash Accounts receivable Fees...
-
Willie Mays, with all-around talent, was one of the greatest baseball players of all time. The numbers of stolen bases by Mays are shown in Table 77 for various years. Let n be the number of stolen...
-
A four-pole, 60-Hz, 24-kV, 650-MVA synchronous generator with a synchronous reactance of 1.82 per unit is operating on a power system which can be represented by a 24-kV infinite bus in series with a...
-
What types of financial policies should the board regulate and what types belong to the staff ?
-
IT, Inc., has adopted the balanced scorecard approach to motivate the managers of its product divisions to work toward the companywide goal of leading its industry in innovation. The corporations...
-
When a public firm has seasoned offerings in the market the stock price generally will __ This is consistent with the production of ___ theory. A. rise; window of opportunity b. fall;signaling...
-
Harry is a trader selling goods on credit. His financial year ends on 31 December. The balances on his books on 1 January 203 included the following: $ Provision for doubtful debts...
-
The tranquilizing drug meprobamate (Equanil or Miltown) can be synthesized from 2-methylpentanal as follows. Give structures for meprobamate and for the intermediates AC: HCHO, HO H [A (C.H,102)]...
-
The tranquilizing drug meprobamate (Equanil or Miltown) can be synthesized from 2-methylpentanal as follows. Give structures for meprobamate and for the intermediates AC: H HCHO, HO [A (C,H,O,)]...
-
The owner of Midwest Grocers Warehouse is considering a $ 195,000 installation of a new refrigerated storage room. The storage room has an expected life of 20 years with no salvage value . The...
-
In car painting the spray gun is charged positively. Therefore paint particles become positively charged. When the paint is sprayed particles repel each other. Also the car is grounded. Grounding...
-
Given the information answer the question. Stock proceeds: $7,500 Debt payment: ($5,000) From the above information: What is the cash flow from financial activities?
-
Two alpha particles (mass = 6.6 x 1027 kg & charge = 3.2 x 10-19 C), separated by an enormous distance, approach each other along a "head-on" collision path. Each has a speed of 3.0 x 10 m/s...
-
The following angles are given in degrees. Convert them to radians: a. 20 b. 35 c. 80 d. 270
-
A college student realized that he was spending too much money on movies. For the remaining 5 months of the year his goal is to spend a mean of $40 a month towards movies. How much can he spend in...
-
Access the September 27, 2014, 10-K report for Apple, Inc. (ticker AAPL), filed on October 27, 2014, from the EDGAR filings at www.SEC.gov. Required 1. What products are manufactured by Apple? 2....
-
Show that, given a maximum flow in a network with m edges, a minimum cut of N can be computed in O(m) time.
-
Limonene, a major component of lemon oil, has the formula C10H16. (a) On reaction with excess H2 in the presence of Pt, limonene produces C10H20. What information does this provide about the...
-
Show the structures of A, B, C, and D in the following reactionsscheme: D Optically inactive H,SO. H,O B Pt C,H14 C,H12 Optically Optically inactive active 1) Hg(O,CCH3)2, H20 2) NaBH4, NAOH...
-
In Figure, suppose Br2 adds to the alkene from the bottom, rather than from the top as shown. Analyze the stereochemistry of the reaction in this case and explain which products areformed. Relative...
-
An individual can win a bouncy ball by guessing under which one of four cups the ball is located. After each guess, if the ball is won, a new ball is placed randomly under one of the four cups. If...
-
Newport Beach, October 1, 2022 After describing the revenue model assumptions to Moore-Cash, Sleepless discussed the assumptions for the remainder of the operating model. "ADS's cost structure...
-
Scott is the sole proprietor of an auto repair business and periodically makes withdrawals from his business account for personal use. How should Scott's bookkeeper label the account in which these...

Study smarter with the SolutionInn App


