Provide a mechanism for the following reaction. NO NO 2 ss-sr NaOCH3 OH NO O=5 S=0 NO
Question:
Provide a mechanism for the following reaction.
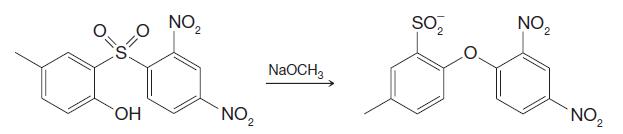
Transcribed Image Text:
NO₂ NO₂ 2 ss-sr NaOCH3 OH NO₂ O=5 S=0 NO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Provide a mechanism for the following reaction and explain why it occurs faster than nitration of benzene. NO2
-
Provide a mechanism for the following reaction OH H2SO4 H20
-
Provide a mechanism for the following reaction. Draw a reaction energy coordinate diagram that illustrates the kinetic and thermodynamic pathways for this reaction. HBr Br+
-
MAX DIVISOR TREE Let a tree exists with the root value N. The property of this tree is that each of its nodes branches out to the nodes with a value equal to one of its divisors (except 1 and the...
-
2x 6 Describe the solution set as an inequality, in interval notation, and on a graph.
-
The following are historical data on staff salaries (dollars per pupil on 30 schools sampled in the eastern part of the United States in the early 1970s).(a) Compute the sample mean and sample...
-
The stockholders' equity of Ranger Corporation at January 1 appears below: During the year, the following transactions occurred: May 12 Declared a 15 percent stock dividend; market value of the...
-
Prepare journal entries to record the following. (a) Sound Tracker Company retires its delivery equipment, which cost $41,000. Accumulated depreciation is also $41,000 on this delivery equipment. No...
-
Project L requires an initial outlay at t = 0 of $ 5 5 , 0 0 0 , its expected cash inflows are $ 8 , 0 0 0 per year for 9 years, and its WACC is 9 % . What is the project's MIRR? Do not round...
-
Consider a CMOS process for which Lmin = 0.25 m, tox = 6 nm, n 460 cm2/V . s, and V; %3| 0.5 V. (a) Find Cox and k', (b) For an NMOS transistor with W/L = 15 um/0.25 um, calculate the values of Voy ,...
-
Predict the major product from each of the following reactions. H O (1) (2) HO+ (3) HCrO4 MgBr (excess)
-
Predict the outcome of the following reactions: CN CI 2 equiv. KNH lig. NH3, 33 C
-
A playground merry-go-round has a mass of 120 kg and a radius of 1.80 m and it is rotating with an angular velocity of 0.500 rev/s. What is its angular velocity after a 22.0-kg child gets onto it by...
-
In this week's reading, I was able to learn that there are lots of different types of advanced directives but the most popular ones are directives to physicians and family (living will), medical...
-
Back in 2003, Nicholas Carr set off a firestorm of controversy in the tech world with his Harvard Business Review article, "IT Doesn't Matter" (OK, maybe "firestorm" is an overstatement, but it was a...
-
Please respond to this discussion post below detailed and throughly, Reducing minimum requirements for law enforcement recruitment can be a double-edged sword. On one side, it could open the door to...
-
Prior to any questioning, the person must be warned that he [or she] has the right to remain silent, that any statement he [or she] does make may be used as evidence against him [or her], and that he...
-
Over a time interval of 1.80 years, the velocity of a planet orbiting a distant star reverses direction, changing from +20.0 km/s to -20.1 km/s. Find (a) the total change in the planet's velocity (in...
-
Suppose a stock had an initial price of $61 per share, paid a dividend of $1.40 per share during the year, and had an ending share price of $69. Compute the percentage total return. What was the...
-
Data on weekday exercise time for 20 females, consistent with summary quantities given in the paper An Ecological Momentary Assessment of the Physical Activity and Sedentary Behaviour Patterns of...
-
Explain why this compound gives about 50%syn elimination whenheated: N(CH3)3
-
When 2-bromo-2-methylbutane is heated in a mixture of ethanol and water, it gives a 64% yield of substitution products and a 36% yield of elimination products. (a) What mechanisms does this reaction...
-
This elimination reaction gives a single product. Show its structure and explain why it is the only productformed. O I CH,CCH CCH,CH, + KOH - ELOH H,CH3
-
Carlyle Corp purchased business equipment in 2020 for $150,000. In 2023 he sold the property for $175,000. At the time of sale the accumulated depreciation was $75,000 and subject to IRC Sec 1245...
-
CASE QUESTIONS 1. What responsibilities, control, and costs does V2 bear under each of the FOB terms offered? 2. What is the delivery cost and landed cost per set of speakers for each delivery...
-
Southern Corporation has a capital structure of 4 0 % debt and 6 0 % common equity. This capital structure is expected not to change. The firm's tax rate is 2 1 % . The firm can issue the following...

Study smarter with the SolutionInn App


