How many signals do you expect in the 13 C NMR spectrum of each of the following
Question:
a.
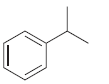
b.
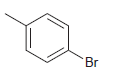
c.
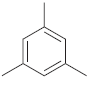
d.
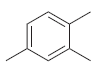
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a 6...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many signals do you expect in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) Geraniol Isolated from roses and used in perfumes . NH2 Dopamine A neurotransmitter that is...
-
Consider the following compound: (a) How many signals do you expect in the 1 H NMR spectrum of this compound? (b) Rank the protons in terms of increasing chemical shift. (c) How many signals do you...
-
Dimethylformamide (DMF) is a common solvent: (a) The 1 H NMR spectrum of DMF exhibits three signals. Upon treatment with excess LAH followed by water, DMF is converted into a new compound that...
-
Solve each system. If a system is inconsistent or has dependent equations, say so. -5x + 2y = -4 6x + 3y = -6
-
Assume the role of a social science researcher in charge of developing a new crime prevention program targeted toward juveniles in the K-12 school system. Review current crime prevention programs...
-
A linear plot of speed versus elapsed time has a slope of 6.0 m/s2 and a vertical intercept of 3.0 m/s. (a) What is the change in speed in the time interval between 4.0 s and 6.0 s? (b) What is the...
-
Tharp Energy Company, which uses the successful efforts method of accounting, owns an individually significant lease, with a cost of $200,000. On December 31, 2017, the lease is not considered...
-
Ingersoll Rand operates as a manufacturer in four segments: Air Conditioning Systems and Services, Climate Control Technologies, Industrial Technologies, and Security Technologies. They installed an...
-
What is the annual growth rate of P(t) = 50(1.62)* (where t is given in years) as a percent? Be sure to show all your work.
-
John Hall tried to unclog a floor drain in the kitchen of the restaurant where he worked. He used a drain cleaner called Zaps Blue Lye that contained crystalline sodium hydroxide (the chemical name...
-
How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy? Problem 15.29 a. b. OH HO. HO m/z = 126.0315 m/z = 126.1404
-
Draw a Frost circle for the following cation, and explain the source of instability of this cation.
-
Describe how CASE is used to support each phase of the SDLC.
-
What is the key problem in the case study Under Armour: Creating and Growing a New Consumer Brand. What are two solutions to the problems. What are advantages and disadvantages.
-
what is the problem faced by retail industry in malaysia? give factors that influence the inventory management operation?
-
Problem 4 Determine the equations of the slope and deflection curves for a beam shown in figure below. Use the Clebsch's method. Data: P, 1, EI. The solution should contain coordinate frames, proper...
-
This is the case of Cabot Oil & Gas Corporation v. Daugherty Petroleum, Inc. Question: please summarize and include the following; Fact: Judgment: Issue: Holding: General Analysis Applied Analysis:...
-
(a) In the Hough Transform, a point (x0, y0) in the xy-plane is mapped into a curve in the (p, 0)-parameter space. Write down the equation of the curve. (b) If we apply the Hough transform on the...
-
Suppose you would like to build a hash table for images, where the key for each image is a thumbnail image of 7575 pixels, with each pixel being one of 256 possible colors. Describe a hash function...
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Which of the following alkenes would yield the same alcohol from either oxymercuration-reduction or hydroboration--oxidation, and which would give different alcohols? Explain. (a) cis-2-butene (b)...
-
Give the products (if any) expected from the treatment of each of the following compounds with ozone followed by dimethyl sulfide (a) (b) 2-methylpentane CH2
-
Give the products (if any) expected when the compounds in Problem 5.13 are treated with ozone followed by aqueous hydrogen peroxide.
-
Question 20 (1 point) Listen The Carters contributed $8,000 to the Path for Progress Political Action Committee What is the tax treatment of this contribution? O Deductible on Schedule E as an...
-
The Sheffield and Frizell Dental Clinic provides both preventive and orthodontic dental services. The two owners, Reese Sheffield and Anita Frizell, operate the clinic as two separate investment...
-
Montoure Company utiliza un sistema de inventario peridico. Realiz las siguientes transacciones de compra y venta en el ao calendario. Fecha Actividades Unidades Adquiridas al Costo Unidades vendidas...

Study smarter with the SolutionInn App


