How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy? Problem 15.29
Question:
How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy?
Problem 15.29
a.
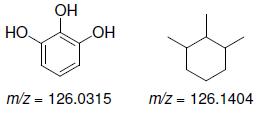
b.
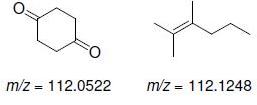
Transcribed Image Text:
OH HO. HO m/z = 126.0315 m/z = 126.1404
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a The first compound should have a very broad signal between 3200 and 3600 cm 1 co...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you distinguish between each pair of compounds using mass spectrometry? a. b. N.
-
How would you distinguish between each pair of compounds using high-resolution mass spectrometry? a. b. .
-
How would you distinguish between the following pairs of compounds using simple chemical tests? Tell what you would do and what you would see. (a) Cyclopentane and cyclopentane (b) 2-Hexene and...
-
If $4000 is deposited into an account paying 3% interest compounded annually and at the same time $2000 is deposited into an account paying 5% interest compounded annually, after how long will the...
-
Here is the article from 'Required Resources': abcnews.go.com/Health/story?id=117304&page=1 Read this article on the dangers of roller coasters. After you've read the article, write a short summary...
-
Carr Corporation is considering new equipment. The equipment can be purchased from an overseas supplier for $3,900. The freight and installation costs for the equipment are $515. If purchased, annual...
-
Question: Dr. Warfield hired Wolfe, a young carpenter, to build his house. A week or so after they signed the contract, Wolfe filed Articles of Incorporation for Wolfe Construction, Inc. Warfield...
-
Financial data for Joel de Paris, Inc., for last year follow: The company paid dividends of $15,000 last year. The Investment in Buisson, S.A., on the balance sheet represents an investment in the...
-
What do you think about the myriad of News Broadcast switching from a more journalistic basis of reporting to the masses, and now seemingly changing to a more gossip-like review?
-
Florida Energy Restoration, Ltd. (FER) enters into a lease agreement on January 1, 2018, to lease standard power generators from R&R Electric, Inc. The terms of the lease follow. The term of the...
-
Rank each of the bonds identified in order of increasing wave number. R-CEN - N-H - -
-
How many signals do you expect in the 13 C NMR spectrum of each of the following compounds? a. b. c. d. Br
-
Compute the following derivatives using the method of your choice. d dx (cos (x sin x))
-
A horizontal pipe of diameter 0.779 m has a smooth constriction to a section of diameter 0.4674 m. The density of oil flowing in the pipe is 821 kg/m. If the pressure in the pipe is 7530 N/m and in...
-
A researcher looking into the impact of gender, age, and the number of hours worked on the income of managers, created a model using the natural log of the individuals wage as the dependent variable....
-
Explain the difference between D-Latch and D flip flop with the help of diagram? If the sand Rwaveforms in Figure 2 are applied to the inputs of the latch as shown, determine the waveform that will...
-
Prepare the following for a report for the company Australia Bay Seafoods to market itsproducts internationally by comparing the opportunities and threats in countries Korea and Japan. Then decide...
-
The objective lens of a microscope has a focal length of 5.0mm. Part A What eyepiece focal length will give the microscope an overall angular magnification of 300? Assume a length L = 160 mm. Express...
-
The Orange County Department of Public Health tests water for contamination due to the presence of E. coli (Escherichia coli) bacteria. To reduce laboratory costs, water samples from six public...
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Iodine azide,I-Nr, adds to isobutylene in the following manner: iodine azide
-
For each of the following cases, provide the structure of an alkene that would give the alcohol as the major (or only) product of hydroboration--oxidation. CH CH CH CH OH CH, CH,
-
Which of the following alkenes would yield the same alcohol from either oxymercuration-reduction or hydroboration--oxidation, and which would give different alcohols? Explain. (a) cis-2-butene (b)...
-
Green LTD has net sales of $1,642,500, a beginning balance of net receivables of $187,500, and an ending balance of net receivables of $235,500 has a days' sales in receivables (rounded) of: Group of...
-
In chapter 35, how are costs associated with ERM implementation taken into account? Question 20 options: A) They are disregarded due to the commitment of the board to ERM B) Costs always drive the...
-
A company's lag indicators are Blank______. Multiple select question. measures of final outcomes of earlier management decisions key financial measures more useful for performance management and...

Study smarter with the SolutionInn App


