Rank each of the bonds identified in order of increasing wave number. R-CEN - N-H - -
Question:
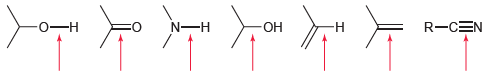
Transcribed Image Text:
R-CEN -о—н N-H -ОН -н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
OH NH In...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank each of the following series of compounds in order of increasing oxidationlevel: CI (a) (b) CH3CN CH3CH2NH2 H2NCH2CH2NH2
-
Rank the following bonds in order of increasing ionic character: NOO, CaOO, COF, BrOBr, KOF.
-
The partial Lewis structure that follows is for a hydrocarbon molecule. In the full Lewis structure, each carbon atom satisfies the octet rule, and there are no unshared electron pairs in the...
-
Write the expression as one ratio without any negative exponents. x1/4x-3/4 X
-
Discuss the growing income inequality in the U.S. and the world with someone mature enough to have some perspective from experience about it. Ask them what they think about the widening gap between...
-
Estimate the number of automobile repair shops in the city you live in by considering its population, how often an automobile needs repairs, and how many cars each shop can service per day. Then look...
-
Rock Petroleum began operations in 2016 with the acquisition of four undeveloped leases, all individually significant. Give the entries assuming the following transactions. For simplicity, you may...
-
Refer to the data in Problem 14-40. R&D is assumed to have a two-year life in the AC Division and a nine-year life in the SO division. All R&D expenditures are spent at the beginning of the year....
-
dy For each of the following curves, use implicit differentiation to find and determine the equation dx of the tangent line at the given point. (a) x3 y3 = 6xy at ( - 3,3) dy da dy dx |(-3,3) =...
-
A 240 mm dia. steam pipe, 200 m long is covered with 50 mm of high-temperature insulation of thermal conductivity 0.092 W/m-K and 50 mm low-temperature insulation of thermal conductivity 0.062 W/m-K....
-
All of the following compounds absorb IR radiation in the range between 1600 and 1850 cm -1 . In each case, identify the specific bond(s) responsible for the absorption(s), and predict the...
-
How would you distinguish between each pair of compounds in Problem 15.29 using IR spectroscopy? Problem 15.29 a. b. OH HO. HO m/z = 126.0315 m/z = 126.1404
-
The quantities of pants, shirts, and jackets owned by Mike and Don are given by the matrix A, and the costs of these items are given by matrix B. (a) Calculate the matrix AB. (b) Interpret the...
-
What is the customer value that DBS is looking to create by using AI? What are DBS's 3 core principles?
-
Hi! I need to do a case brief for my Business Law class and am stuck on how to complete... I have a very hard time with this class and need help to complete it :( Please see attached for the details,...
-
What is the fundamental challenge buyers face when purchasing under all unit discounts, compared to when they purchase under incremental discounts?
-
What is a current problem effecting the Aviation Industry safety? what are solutions currently in Work, and what do you think is the best solution
-
The fact that restrictive covenants are widely used does not mean, as NanoMech, Inc. v. Suresh shows, that they are always enforceable. NanoMech, Inc. v. Suresh 777 F.3d 1020 (8th Cir. 2015) OPINION...
-
Imagine that you work for an insurance company that is insuring people against identity theft. You have just learned about a major security breach at a prominent bank used by many of your customers....
-
White Bolder Investments (WBI) You are an intern working for WBI, a large investment advisory services in Sydney. Among other regular customers, WBI has been providing advisory services for Jumbo...
-
The industrial synthesis of methyl tert-butyl ether involves treatment of 2-methylpropene with methanol (CH3OH) in the presence of an acid catalyst, as shown in the following equation. CH3 H3C H3C...
-
The standard free energy of activation (G++) for hydration of 2-methylpropene to 2-methyl-2-propanol (Eq. 4.41, p. 169) is 91.3 kJ mol-t (Zt 8 kcal mol-l;. The standard free energy G for hydration of...
-
The standard free energy of activation (G++) for hydration of 2-methylpropene to 2-methyl-2-propanol (Eq. 4.41, p. 169) is 91.3 kJ mol-t (Zt 8 kcal mol-l;. The standard free energy G for hydration of...
-
The comparative statements of Oriole Company are presented here: ORIOLE COMPANY Income Statements For the Years Ended December 31 2022 2021 Net sales $1,895,640 $1,755,600 Cost of goods sold...
-
Lars Linken opened Pronghorn Cleaners on March 1, 2022. During March, the following transactions were completed. Mar Issued 9,800 shares of common stock for $14,700 cash. 1 1 1 2 3 9 6. Borrowed...
-
Outback Outfitters sells recreational equipment. One of the company's products, a small camp stove, sells for $90 per unit. Variable expenses are $63 per stove, and fixed expenses associated with the...

Study smarter with the SolutionInn App


