In the mass spectrum of bromobenzene (Figure 15.27), the base peak appears at m/z = 77. Figure
Question:
Figure 15.27
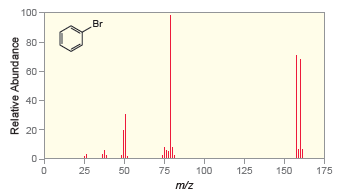
a) Does this fragment contain Br? Explain your reasoning.
b) Draw the cationic fragment that represents the base peak.
Transcribed Image Text:
100 Br 80 60 40- 20 50 25 75 100 125 150 175 m/z Relative Abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (17 reviews)
a This fragment is ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all four resonance forms of the fragment at m/z 73 in the mass spectrum of pentanoic acid.
-
The mass spectrum of 1-butanol shows an intense daughter ion peak at m/z 5 31. Explain how this peak might arise.
-
Identify the expected base peak in the mass spectrum of 2, 2, 3-trimethylbutane. Draw the fragment associated with this peak, and explain why the base peak results from this fragment.
-
As speed increases for an object in free fall, does acceleration increase also?
-
What is the role of specialists and traders on the New York Stock Exchange?
-
According to Denison's estimates, which factors were most important in accounting for the growth in real output over the 1929-82 period?
-
A company maintains its fixed assets at cost. Depreciation provision accounts for each asset are kept. The following additions were made during the financial year ended 31 December 19X9: Machinery...
-
Assume the Bullock Corporation had the following purchases and sales of its single product during its first three years of operation. Cost of goods sold is Bullock's only expense. The income tax rate...
-
introduction to corporate finance fin 430, which is the answer. 12. The Great Empire offers a common stock that pays an annual dividend of RM2 a share. The company has promised to maintain a constant...
-
The 13 C nucleus is a spin 1/2 particle as is a proton. However, the energy splitting for a given field strength is roughly 1/4 of that for a proton. Using a 1.45-T magnet as in Example Problem 30.6,...
-
Show how you would use a Grignard reaction to prepare each compound below. a. b. c. d. e. f. OH
-
Suppose that the pen-making industry is perfectly competitive. Also suppose that each current firm and any potential firms that might enter the industry all have identical cost curves, with minimum...
-
Do you agree with the U.S. governments policies restricting the export of dual-use goods? Why or why not? (You may wish to check out the Bureau of Industry and Securitys website, which details how...
-
What are the similarities and differences between VCA and other forms of data analytics?
-
What is the biggest organization to have compromised the security and privacy of customer or employee data? What role could accountants have played in preventing this breach?
-
What are the top four drivers of learning efficiency?
-
What organizational capabilities do organizations need in order to become true AI companies?
-
Assume that Boardmaster sold skateboards to a department store for \($35,000\) cash. How would this transaction affect Boardmasters accounting equation? a. Increase both assets and stockholders...
-
The Internet is made up of a large number of networks. Their arrangement determines the topology of the Internet. A considerable amount of information about the Internet topology is available on...
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
Draw a Newman projection along the C2-C3 bond of the following conformation of 2, 3-dimethylbutane, and calculate total strainenergy:
-
Identify the functional groups in the following substances, and convert each drawing into a molecular formula (red = O, blue =N): (a) (b) Phenylalanine Lidocaine
-
Give IUPAC names for the following alkanes, and convert each drawing into a skeletal structure: (a) (b) (c) (d)
-
Tubaugh Corporation has two major business segments-East and West. In December, the East business segment had sales revenues of $410,000, variable expenses of $220,000, and traceable fixed expenses...
-
Assume that actual overhead costs were $77,000 and overhead allocated to jobs was $53,000. The unadjusted balance in Manufacturing Overhead would be because the overhead was OA. $24,000 credit...
-
4. The city of Alpine incurred the following costs during the year in its property tax collection department: Purchase of computer equipment Salaries and wages Purchase of electricity from the...

Study smarter with the SolutionInn App


