Show how you would use a Grignard reaction to prepare each compound below. a. b. c. d.
Question:
Show how you would use a Grignard reaction to prepare each compound below.
a.
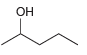
b.
![]()
c.
![]()
d.
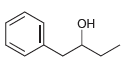
e.
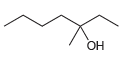
f.
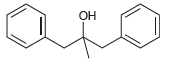
Transcribed Image Text:
OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a b c d e ...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use a Grignard reaction to prepare the following alcohols: (a) 2-Methyl-2-propanol (b) 1-Methylcyclohexanol (c) 3-Methyl-3-pentanol (d) 2-Phenyl-2-butanol (e) Benzyl alcohol (f) 4-Methyl-1-pentanol
-
Show how you would use a Suzuki reaction to synthesize Bombykol, the sex hormone of the silk moth, from cis-1- bromopent-1-ene and the acetylenic alcohol shown below. Bombykol
-
Show how you would use a Suzuki reaction to synthesize the following biaryl compound. As starting materials you may use the two indicated compounds, plus any additional reagents you need. Make OCH3...
-
Harley-Davidson: Preparing for the Next Century There are very few products that are so exciting that people will tattoo your logo on their body. Richard Teerlink, Retired CEO, Harley-Davidson In...
-
Why are stock market analysts so concerned about what's going on at the Federal Reserve?
-
The pH of a bicarbonate-carbonic acid buffer is 8.00. Calculate the ratio of the concentration of carbonic acid (H2CO3) to that of the bicarbonate ion (HCO3-).
-
A department of Calcutta Textiles produces cotton fabric. All direct materials are introduced at the start of the process. Conversion costs are incurred uniformly throughout the process. In April...
-
A chain of sport shops catering to beginning skiers, headquartered in Aspen, Colorado, plans to conduct a study of how much a beginning skier spends on his or her initial purchase of equipment and...
-
MCV4U-Course Culminating Task Final Course Culminating Task (C.C.T.; 20% of the Final Marks) Task 1: The Elasticity of Demand Ideas and Questions: Read the information given for each task and take...
-
Draw the developed diagram of a 2-layer simplex progressive lap- winding for a 2-pole DC generator with 8 coils. Then, find the positions of the brushes on the commutator segments
-
Draw a bond-line structure for each of the following compounds: a. 3-Isopropyl-2, 4-dimethyl-2-pentene b. 4-Ethyl-2-methyl-2-hexene c. 1, 2-Dimethylcyclobutene (The name of a cycloalkene will not...
-
In the mass spectrum of bromobenzene (Figure 15.27), the base peak appears at m/z = 77. Figure 15.27 a) Does this fragment contain Br? Explain your reasoning. b) Draw the cationic fragment that...
-
In sales promotion planning, what is meant by logistics and fulfillment?
-
Given the below objectives. Formulate the following giving more details referring to the literature Background of the research Research gap Problem statement Research objectives; To investigate the...
-
In 2024, the Westgate Construction Company entered into a contract to construct a road for Santa Clara County for $10,000,000. The road was completed in 2026. Information related to the contract is...
-
Colonial Pharmaceuticals is a small firm specializing in new products. It is organized into two divisions, which are based on the products they produce. AC Division is smaller and the life of the...
-
Cruz Corporation has $50 billion of debt outstanding. An otherwise identical firm has no debt and has a market value of $150 billion. Under the Miller model, what is Cruz's value if the...
-
5. Your next exercise is to write a program to read a sequence of values and add them to an array. You will then find the largest and the smallest values of the array, print them to the screen, and...
-
Marcus, a cash basis individual, is a general partner in MNOP Partnership. Both Marcus and MNOP use a calendar year for tax purposes. According to the partnership agreement, MNOP pays a $10,000...
-
What are technical skills At what level are they most important and why?
-
Make a graph of potential energy versus angle of bond rotation for propane, and assign values to the energy maxima.
-
Consider 2-methylpropane (isobutene). Sighting along the C2-C1 bond: (a) Draw a Newman projection of the most stable conformation. (b) Draw a Newman projection of the least stable conformation. (c)...
-
Sight along the C2-C3 bond of 2, 3-dimethylbutane, and draw a Newman projection of the most stable conformation.
-
Question: The Principle Limited Partnership has more than 300 partners and is publicly traded. The Principle was grandfathered under the 1987 Tax Act and has consistently been taxed as a partnership....
-
Wicker Corporation made estimated tax payments of $6000 in Year 1. On March 12 of Year 2, it filed its Year 1 tax return showing a $20,000 tax liability, and it paid the $14,000 balance at the time....
-
Assume you are wrapping up the 20X4 audit for your client, West Tech Inc. (WT), a manufacturer of cell phones, tablets, and personal computers. To date there has been every indication that the...

Study smarter with the SolutionInn App


