Using cyclopentanone as your starting material and using any other reagents of your choice, propose an efficient
Question:
(a)
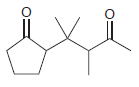
(b)
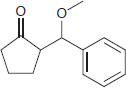
(c)
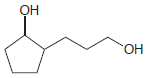
Transcribed Image Text:
OH HO.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a b ...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose an efficient synthesis for each of the following compounds using the acetoacetic ester synthesis. (a) (b) (c) (d)
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH H.
-
(a) Decide whether each of the following expressions are true or not. Answer yes or no. In any case where it is not true, provide the actual O-complexity. (i) log n = O(n!) (ii) 10! = O(log n) (iii)...
-
$1,000 is expected to be received 10 years from now. The present value of this $1,000 future cash flow is: 1: Always greater than $1,000. 2: Greater if the interest rate goes up. 3: Less than $1,000...
-
Provide no-arbitrage arguments for equation (6.9).
-
Suppose that you just turned 30 and are planning for your retirement. You hope to retire at the age of 67, and would like to be able to make end-of-month withdrawals from your retirement account of...
-
Because of the individuality of people, there always exist differing views of what management is all about. Below are lists of possible perspectives and a selected group of organizational members....
-
The time required for workers to produce each unit of a product decreases as the workers become more familiar with the production procedure. It is determined that the unction for the learning process...
-
A hospital in a developing nation is forced to manually put the flu shot vaccine into syringes. The recommended dosage for the flue shot is 0.5mL. The local health authorities define all syringes...
-
Identify the reagents you would use to prepare the following compound via a Robinson annulation. . 0=
-
Refer to the illustrative example of Chapter 7 where we fitted the Cobb Douglas production function to the manufacturing sector of all 50 states and the District of Columbia for 2005. The results of...
-
Under standard accounting rules, it is possible for a companys liabilities to exceed its assets. When this occurs, the owners equity is negative. Can this happen with market values? Why or why not?
-
Cash balances often constitute a very small percentage of an entity's current or total assets. Why then isn't less effort allocated to auditing cash balances?
-
What special considerations relate to applying the concepts of materiality and preliminary audit strategies to the audit of cash balances?
-
What could you do with 2G that was not possible with 1G?
-
What can you do with 5G that you cant do with 4G?
-
a. How and by whom is the opinion on the financial statements formulated, and how is it communicated? b. How are proposed adjustments and disclosures generally resolved?
-
In 2008, the Labour Party in Britain promised that patients would have to wait for no more than four hours to be seen in an emergency room. The National Health Service started rewarding hospitals...
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
Propose a structure for a compound A that has an infrared absorption at 1820 cm-1 and a single proton NMR absorption at 1.5. Compound A reacts with water to give dimethylmalonic acid and with...
-
Pentanoic acid and methyl butyrate are constitutional isomers. Which has the higher boiling point and why?
-
Klutz McFingers. a graduate student in his ninth year of study, has suggested the following synthetic procedures and has come to you in the hope that you can explain why none of them works very well...
-
31 BT E R ole]1i 1o 2111Trial balancev For the Quarter Ended May 31, 2022For the Year Ended May 31, 2022May 31, 2022 Totals $ $ Current Attempt in Progress Julia Dumars is a licensed CPA. During the...
-
Chief Customer Officer Jayden Hayden has asked sales analyst Paige Sage to analyze the company's Western Kentucky district. Paige intends to calculate ROAM for the district. She has compiled the...
-
Which of the following statements is correct?If the demand curve for a firm's stock is relatively flat, the firm will have a more difficult time raising alarge amount of new equity funds for...

Study smarter with the SolutionInn App


