The molecules in a six-particle gas have velocities Calculate (a) v avg , (b) v avg ,
Question:
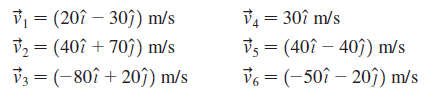
Calculate
(a) vavg,
(b) vavg,
(c) vrms.
Transcribed Image Text:
V1 = (20î – 30) m/s V2 = (40î + 70î) m/s V3 = (-80î + 20ĵ) m/s V = 30î m/s Vs = (40î – 407) m/s 36 = (-50î – 203) m/s %3D %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
Solve a In tabular form we have Particle v x ms v y ms ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
In our discussions of kinetic theory, we have focused on the motion of the molecules in a gas. The result for the typical speed in Equation 15.18, however, also applies to molecules in a liquid. Use...
-
We have two equal-size boxes, A and B. Each box contains gas that behaves as an ideal gas. We insert a thermometer into each box and find that the gas in box A is at a temperature of 50C while the...
-
Why wouldn't you expect all the molecules in a gas to have the same speed?
-
Using the aggregate expenditures table below, answer the questions that follow. a. Compute the APC when income equals $2,300 and the APS when income equals $2,800. b. Compute the MPC and MPS. c. What...
-
Which of the following individuals qualify for the earned income credit for 2016? a. Thomas is single, is 21 years of age, and has no qualifying children. His income consists of $9,000 in wages. b....
-
During your audit of the financial statements of the Dolomite Corporation for the year ended December 31, 19X1, you determine that the company's surplus funds have been temporarily invested in...
-
The 2008 comparative balance sheet and income statement of Digital Subscriptions, Inc.. follow. Digital Subscriptions had no noncash investing and financing trans- actions during 2008. During the...
-
Which of the following observations appear to indicate market inefficiency? Explain whether the observation appears to contradict the weak, semi-strong, or strong form of the efficient-market...
-
16.Fifteen people work in an office. 9 are women and 6 are men.The flu virus is coming. (a)In how many ways can the flu virus randomly select 7 workers out of the 15 to get sick?Show work. (b)In how...
-
Magic Enterprises produces three perfumes. The sales department prepared the following tentative sales budget for the first quarter of the coming year: Perfume Units Moon Glow ............. 250,000...
-
The two containers of gas in FIGURE Q20.8 are in good thermal contact with each other but well insulated from the environment. They have been in contact for a long time and are in thermal...
-
Suppose you place an ice cube in a beaker of room temperature water, then seal them in a rigid, well-insulated container. No energy can enter or leave the container. a. If you open the container an...
-
Refer to Cornerstone Exercises 2.2 and 2.3. Next year, Pietro expects to produce 50,000 units and sell 49,300 units at a price of $12.50 each. Beginning inventory of finished goods is $42,500, and...
-
Discuss the role of linear programming in project management and how it can be used to optimize resource allocation and scheduling.
-
The function f(x) = 2x - x2 is given graphed below: Note: Click on graph for larger version in new browser window. (A) Starting with the formula for f(x), find a formula for g(x), which is graphed...
-
OA solid object has an apparent weight 15.2 N when completely submerged in ethyl alcohol (density 790) and 13.7 N when completely submerged in water. Find the volume of the object.
-
What is Business Management? Business Management is the process of developing and executing a plan to achieve objectives and success in an organization.
-
A diver leaves a 3-m board on a trajectory that takes her 2.2m above the board and then into the water 2.6m horizontally from the end of the board. a) At what speed did she leave the board? Express...
-
Wegener proposed that the worlds continents had at one time all been joined together into one supercontinent. What name is given to this supercontinent?
-
1. Which of the four major types of information systems do you think is the most valuable to an organization? 2. How do you critically associate the ideas of business agility and business efficiency...
-
Which isotope of lead is commonly found within uranium ore deposits.
-
What happens to the uranium-235 nucleus when it is stretched out?
-
What three fissionable isotopes work well in the production of energy within a nuclear power plant?
-
The human body has a blood volume of about 5 liters (p = 1060 kg / m3). The total volume of blood with a cell density of 1225 kg / m3 turned out to contain 1% white blood cells and 44% red blood...
-
Matt is a 23 year old male athlete training for a marathon. Heis also trying to lose weight. His height is 511, weight is 235lbs., usual weight is 252 lbs. He is currently doing a low-carbdiet that...
-
The Sports Equipment Division of Harrington Company is operated as a profit center. Sales for the division were budgeted for 2020 at $896,000. The only variable costs budgeted for the division were...

Study smarter with the SolutionInn App


