Where would Kb emission peaks for Ti, Se, and Zr be found in Figure 21-31? Why are
Question:
Where would Kb emission peaks for Ti, Se, and Zr be found in Figure 21-31? Why are they not labeled?
In Figure 21-31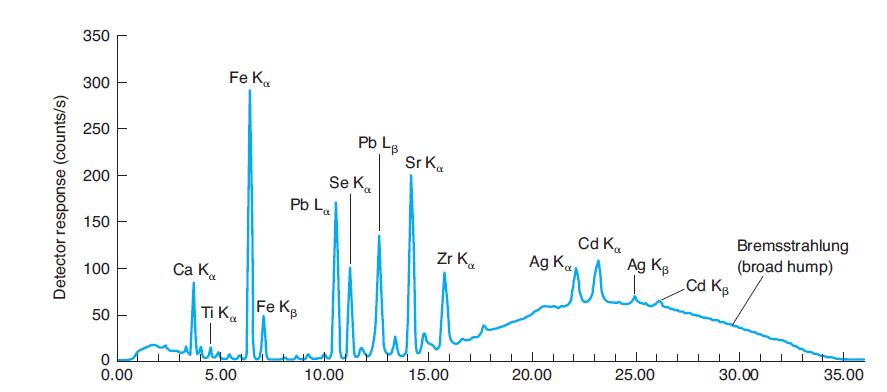
Transcribed Image Text:
350 Fe K. 300 250 Pb L. Sr K. Se K. Pb La 200 150 Cd K. Ag K Bremsstrahlung (broad hump) Cd Kg Zr K. Ag Kg 100 Ca K. Ti Ka Fe KB 50 25.00 30.00 35.00 5.00 10.00 15.00 20.00 0.00 Detector response (counts/s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
For Ti Kbeta line is at 493181kev so next to the ka...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What is a hedge fund? Why are they not a recommended investment for most investors?
-
Why are higher resolution monochromators found in ICP atomic emission spectrometers than in flame atomic absorption spectrometers?
-
Why are La and Lb peaks, but not Ka and Kb peaks for lead identified in Figure 21-31? Why are La and Lb peaks not identified for iron in Figure 21-31? In Figure 21-31 350 Fe K. 300 250 Pb L. Sr K. Se...
-
Medallion and RIEF (Renaissance Institutional Equity Fund) are both managed by Renaissance Technologies. How do they differ in terms of asset classes, dollar capacity, average holding period of each...
-
What is an organization's responsibility in regard to sexual harassment among coworkers or supervisor subordinate pairs? Do you think that Daryl Kolendich responded appropriately to the problem?
-
(a) Given the product structure and master production schedule (Figure 14.14 below), develop a gross requirements plan for all items. (b) Given the preceding product structure, master production...
-
Which word best describes an organizational culture that embraces multiculturalism and in which workforce diversity is highly valued? (a) inclusion (b) effectiveness (c) dynamism (d) predictability
-
At December 1, 2014, Imalda Inc. reported the following information on its statement of financial position: Trade receivables ................. $ 154,000 Allowance for doubtful accounts...
-
Majestic Corporation began the year with accounts receivable, inventory, and prepaid expenses totaling $67,000. At the end of the year, Majestic had a total of $77,000 for these current assets. At...
-
When t = 0, the train has a speed of 8 m/s, which is increasing at 0.5 m/s 2 . Determine the magnitude of the acceleration of the engine when it reaches point A, at t = 20 s. Here the radius of...
-
Explain why X-ray fluorescence is observed when matter absorbs X-rays of sufficient energy. Why does each element have a unique X-ray signature?
-
How much energy in kJ/mol is released when nitrogen emits Ka radiation at 0.392 keV? Compare the Ka energy to 945 kJ/mol, which is the energy required to break the triple bond in N 2 (one of the...
-
Match each of the key terms with the definition that best fits it. ______________ A systems development approach that originated in northern Europe, in which users and the improvement of their work...
-
Explain the key provisions of the Fair Work Act 2009. Explain the purpose of the National Employment Standards and the 10 minimum entitlements. Explain the use of modern awards in workplaces....
-
Describe three character traits of the character Sindbad. Provide examples of the traits with citation.
-
What code(s) may be reported for prolonged services that are neither face-to-face time in the outpatient, inpatient, or observation setting nor additional unit/floor time in a facility or observation...
-
University students have many competing demands that can lead to feeling stressed or overloaded. At the same time, they often face these demands away from their usual supports of family and friends,...
-
Of the key factors the logistics manager can contribute to in the planning process, which one is not included?
-
What is carriage inwards?
-
Bonus shares can be issued out of revenue reserves. True/False?
-
The diprotic acid H2A has pK1 = 4.00 and pK2 = 8.00. (a) At what pH is [H2A] = [HA-]? (b) At what pH is [HA-] = [A2-]? (c) Which is the principal species at pH 2.00: H2A, HA-, or A2-? (d) Which is...
-
The base B has pKb = 5.00. (a) What is the value of pKa for the acid BH+? (b) At what pH is [BH+] = [B]? (c) Which is the principal species at pH 7.00: B or BH+? (d) What is the quotient [B]/[BH+] at...
-
The acid HA has pKa = 4.00. Use Equations 9-17 and 9-18 to find the fraction in the form HA and the fraction in the form A- at pH = 5.00. Does your answer agree with what you expect for the quotient...
-
Harley has two lines of businessa manufacturing/retail operation and a finance subsidiary. Harley's dealers are independent of Harley and sell motorcycles and related products (e.g. Harley apparel)....
-
Autumn Manufacturing mass produces its product in three departments. A cost analysis for Department 1, the first department in the manufacturing cycle for the month of April, can be found in the...
-
Based on the? accounts, create the journal entries that resulted from the six transactions. Include explanations with each journal entry. Cash Supplies Building Equipment (1) 35,000 (4) 10,000 (2)...

Study smarter with the SolutionInn App


