Why are La and Lb peaks, but not Ka and Kb peaks for lead identified in Figure
Question:
Why are La and Lb peaks, but not Ka and Kb peaks for lead identified in Figure 21-31? Why are La and Lb peaks not identified for iron in Figure 21-31?
In Figure 21-31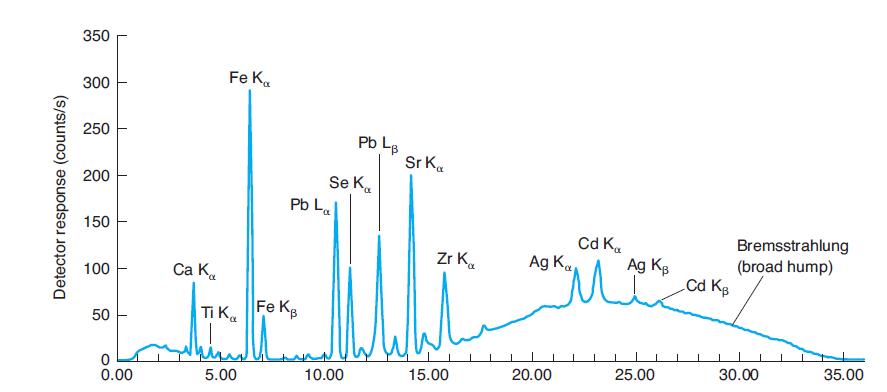
Transcribed Image Text:
350 Fe K. 300 250 Pb L. Sr K. Se K. Pb La 200 150 Cd K. Ag K Bremsstrahlung (broad hump) Cd Kg Zr K. Ag Kg 100 Ca K. Ti Ka Fe KB 50 25.00 30.00 35.00 5.00 10.00 15.00 20.00 0.00 Detector response (counts/s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Answer There are four fundamental processes that will p...View the full answer

Answered By

Enock Oduor
I am a chemist by profession, i coach high school students with their homework, i also do more research during my free time, i attend educational and science fair seminars where i meet students and do some projects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why will a block of iron float in mercury but sink in water?
-
Why do maximizing EPS and maximizing value not necessarily lead to the same conclusion about the optimal capital structure?
-
Why are the multiplet splittings in Figure 28.9 not dependent on the static magnetic field? J12 J42 (yB/2m)(01- 2) Frequency Intensity -----
-
Wollongong Group Ltd, of New South Wales, Australia, acquired its factory building about 10 years ago. For several years the company has rented out a small annex attached to the rear of the building....
-
If you were Erin Dempsey, what would you do?
-
Explain and define Pattern.
-
U.S. Airways recognizes revenue from airline ticket sales when a customer takes a flight or not later than one year from the date of sale (flight coupons have a useful life of one year from the date...
-
My-Best Weight Co. offers personal weight reduction consulting services to individuals. After all the accounts have been closed on November 30, 2012, the end of the current fiscal year, the balances...
-
A farmer gathers 1,200 oranges per week. Based on two samples of 100 oranges, the farmer determines that 88% of the oranges will be of good enough quality to sell to a grocery store with a 4% margin...
-
Sanyu Sony started a new business and completed these transactions during December. Dec. 1 Sanyu Sony transferred $65,000 cash from a personal savings account to a checking account in the name of...
-
Where would Kb emission peaks for Ti, Se, and Zr be found in Figure 21-31? Why are they not labeled? In Figure 21-31 350 Fe K. 300 250 Pb L. Sr K. Se K. Pb La 200 150 Cd K. Ag K Bremsstrahlung (broad...
-
How much energy in kJ/mol is released when nitrogen emits Ka radiation at 0.392 keV? Compare the Ka energy to 945 kJ/mol, which is the energy required to break the triple bond in N 2 (one of the...
-
Carson Chill, Inc., makes ice cream that it sells in 5-gallon containers to retail ice cream parlors. During 2014, the company planned to make 100,000 containers of ice cream. It actually produced...
-
Genetics Engineering is considering the purchase of some new equipment that will cost $300,000 installed. The equipment will produce a product that must be FDA approved and this will require at least...
-
6 Athletic Performance Company (APC) was incorporated as a private company. The company's accounts included the following at July 1: Accounts Payable Buildings. Cash Common Stock $ 4,000 200,000...
-
Lucy has AGI of $125,000 before considering losses from some rental real estate she owns (she actively participates). She had the following losses from her rental property: Rental property 1 Rental...
-
1.1 Distinguish between four levels of strategy in business organisations with specific reference to the decision-making roles of managers. Next to each manager, provide an example of a decision that...
-
S = $1.60/ $ = 2% F= $1.54/E F = 4% Profits from covered interest arbitrage per unit of currency invested are closest to: O A 1.5692 OB. 0.0197 12 OC. $0.0197 OD. $1.5692
-
If, in Example 6.20, the true-false exam had 25 questions instead of 10, which normal curve would you use to approximate probabilities for the number of correct guesses?
-
What are the four types of poultry production systems? Explain each type.
-
A dibasic compound, B, has pKb1 = 4.00 and pKb2 = 6.00. Find the fraction in the form BH2 2+ at pH 7.00, using Equation 9 - 19. Note that K1 and K2 in Equation 9-19 are acid dissociation constants...
-
Write the chemical reactions whose equilibrium constants are Kb1 and Kb2 for the amino acid proline. Find the values of Kb1 and Kb2.
-
What fraction of ethane-1,2-dithiol is in each form (H2A, HA-, A2-) at pH 8.00? at pH 10.00?
-
This is what I need but with these variables: Data Collected ( in dollars ) Like Games Our Play Industry Average Accounts receivable 5 , 4 0 0 7 , 8 0 0 7 , 7 0 0 Net fixed assets 1 1 0 , 0 0 0 1 6 0...
-
Organisational procedures state that all corporate bookings made through SJ Travel incur a 10% mark up to increase profit margins. Show the calculation to mark up a gross fare of $1000.00 by 10% to...
-
all the below calculations should be shown on excel spreadsheet

Study smarter with the SolutionInn App


